Before you read this, I suggest you read post 19.20.
In post 22.13, we saw that we couldn’t use a microscope to find the positions of atoms in molecules. But, in principle, we should be able to obtain some information from an x-ray diffraction pattern. This post gives more details.
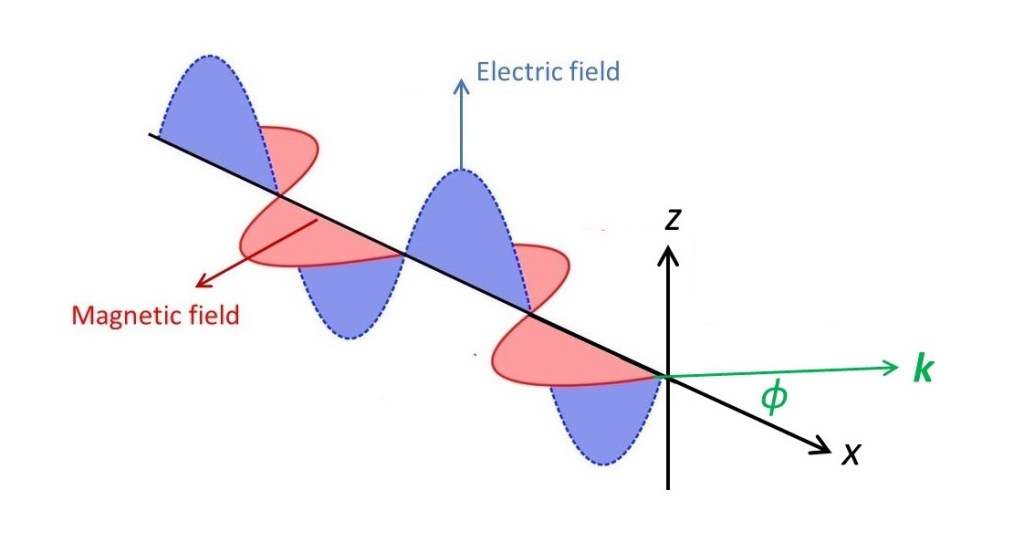
X-rays are scattered by the electrons in matter. Why? Remember that x-rays are electromagnetic waves – they are an oscillating electric field, represented by an electric vector with an oscillating modulus. There is also an oscillating magnetic vector, at right angles to the electric vector and the direction of propagation of the x-rays. But, as the magnetic field is a consequence of the oscillating electric field, we shouldn’t think of them separately – as explained in post 19.9. Let’s think of an x-ray, polarised in the xz plane, in the picture above, where x is in its direction of propagation, incident on an electron at the origin, O, of a Cartesian coordinate system. The electric field (when it is non-zero) repels the electron (because it has a negative electrical charge). So the electron moves up and down in the oscillating electric field of the x-ray wave – like an object floating in a water wave (except that the electron goes down when the wave goes up). In post 19.9, we saw that an oscillating charge produces an electromagnetic wave. So, as the negatively charged electron moves up and down, it acts as a source of an electromagnetic wave with the same frequency as the incident x-ray. The result is that the electron scatters the x-ray. This effect is called Thomson scattering after J J Thomson, the British physicist who discovered the electron.
Note that the x-ray wave is scattered without a change in frequency because the electron moves up and down with the same frequency as the incident x-ray. If we think of the x-ray as a beam of photons, this means that the photons are scattered without any change in momentum (see post 17.30). So Thomson scattering is elastic scattering of x-ray photons. In principle x-ray photons could be scattered by inelastic collision (this is called Compton scattering) or they could lose energy by the photoelectric effect. To investigate the positions of atoms in molecules, we need to use x-rays with a wavelength of about 0.1 nm (see post 22.13). X-rays with this wavelength have a low photon energy so Compton scattering and photoelectric effects are negligible.
Now let’s think about x-rays scattered by the object shown in the picture below that contains many electrons. We will assume that the x-ray waves all have the same frequency and, therefore, the same wavelength. There will be interference between x-rays scattered, in the same direction, by different parts of the object. So, we will see scattering with interference – the definition of diffraction. Many books give the impression that the object must be a crystal but, in this and subsequent posts, we shall see that this impression is false.
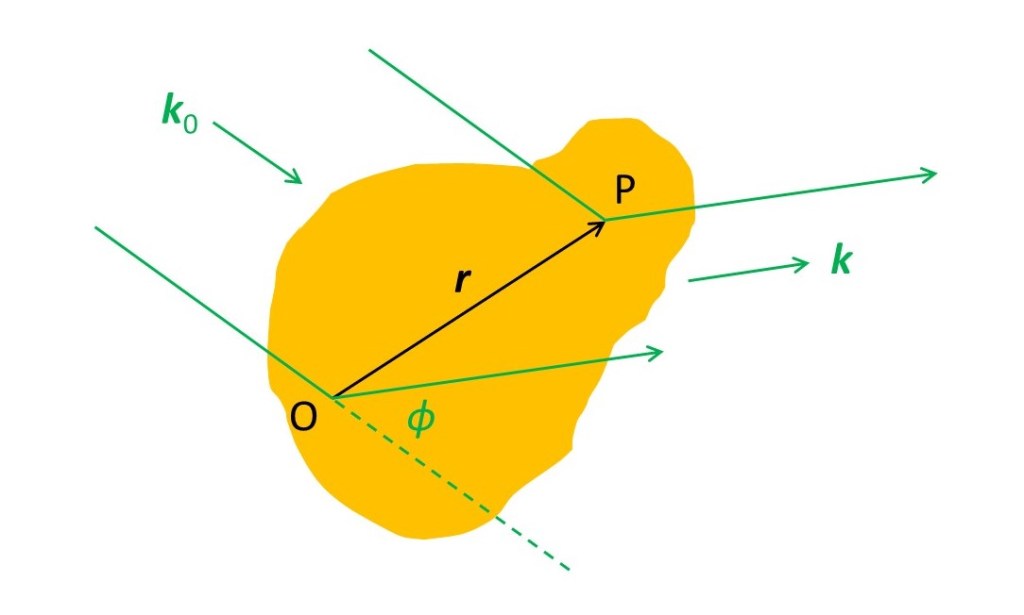
In the picture above the three-dimensional unit vectors k0 and k are in the incident and a scattering direction for the x-rays; k makes an angle φ with k0 in the plane that contains the direction of propagation for the scattered x-rays. We are simply extending the ideas of post 19.20 into three dimensions. Now we are going to consider x-rays scattered by an infinitessimally small volume at P whose position is defined, relative to an origin O in the object by the three-dimensional vector r. We will represent the number of electrons between r and r + δr by ρ(r)δr; ρ(r) is conventionally named the electron density. Note that we often write the electron density as ρ(r) to show that ρ is a function of r – the value of ρ depends on r and may not be the same in different parts of the object. The amplitude of the x-rays scattered by the volume at P is then proportional to ρ(r)δr. The phase of the x-rays scattered by this volume, relative to the phase of x-rays scattered by O is r.K, (the dot product of r and K) as shown in post 19.20 , where
K = (2π/λ)(k – k0)
where λ is the wavelength of the x-rays. Then we can represent the wave scattered by the volume at P, in amplitude and phase by
δψ(K) = ρ(r)δreir.K
as described in post 18.17 where i is the square root of minus 1 and r.K is the dot product of the two vectors. Note that we can write this wave function as δψ(K) to show that δψ is a function of K – its value is different in different scattering directions. Because the amplitude was proportional to ρ(r)δr (so not equal to it) we have missed out a constant from this equation but, as we shall see later, this isn’t usually a problem.
We can find the resultant wave scattered by the object, in the direction of K, by integration giving
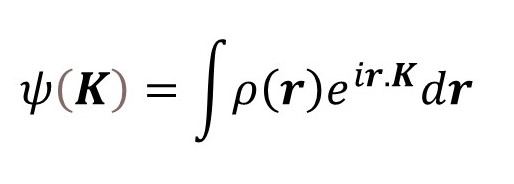
where the limits of integration are over the volume of the object (see post 17.19). Notice that ψ(K) is the Fourier transform of ρ(r). Then, according to post 22.12 we can, in principle, calculate ρ(r) from the inverse transform.
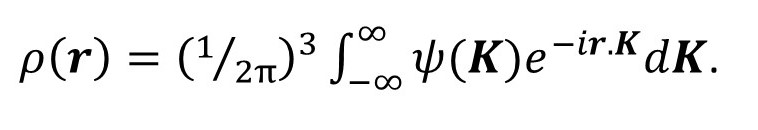
Now (1/2π) is cubed because the Fourier transform is three-dimensional.
But we can’t simply calculate ρ(r) from ψ(K). Why not? Because when we detect the scattered x-ray waves, we measure their energy which depends on ψ2 (see post 19.8) which is equal to ψψ*, where ψ* is the complex conjugate of ψ, so that all the phase associated with the wave is lost – we know nothing about r.K. This problem is called the phase problem of x-ray diffraction analysis.
Also, we don’t measure the absolute energy of the waves scattered in different directions. We only measure their relative energies – so the missing constant, mentioned above, doesn’t affect how we process our data.
In x-ray fibre diffraction, it is conventional to solve the phase problem by molecular model building. If out scattering object is a three-dimensional crystal there are other methods that I hope to explain in later posts.
Related posts
22.13 Resolution of the microscope
22.12 Diffraction…
21.7 The alpha-helix
19.20 Diffraction
19.9 Electromagnetic waves
Follow-up posts
23.04 X-ray crystallography
23.03 X-ray diffraction by a crystal
23.01 Observing x-ray diffraction by a crystal
22.15 X-ray scattering by an atom