Before you read this, I suggest you read post 22.4.
The electrons in an atom can scatter x-rays because they move up and down as the electric vector of the x-rays oscillates, as describe in post 22.4. So, the moving electron is a simple harmonic oscillator if the x-ray wave is a sine wave. The electrons move as the electric vector oscillates because they have an electrical charge. But the protons in an atom also have an electrical charge – so, do they scatter x-rays too? The answer is that scattering by protons in negligible. Why? Because protons have a much higher mass than electrons, so their acceleration is very small in the field represented by the electric vector. Their accelerating charge is the source of the scattered electromagnetic radiation (see post 19.9). Since the acceleration of protons is very small, the intensity of the scattered x-rays is very low.
In this post, I am going to think of an atom as a sphere in which the electron distribution is a function of the distance from the centroid of the nucleus but is independent of direction.
We can say that the assumption I am making is that is that the electron distribution is spherically symmetrical. This is not true because not all atomic orbitals are spherically symmetric. But we usually have no means of fixing the orientation of an atom, so it could be anything. Then our assumption becomes that we can average the electron distribution over all possible orientations of the atom – we can say that the electron distribution has been spherically averaged.
In general, the x-rays scattered by an object can be represented in amplitude and phase by
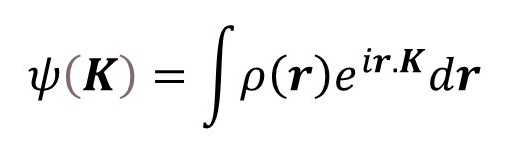
where i is the square root of minus 1, the scattering vector K represents the direction of the scattered x-rays and ρ(r) is the electron density as a function of r, a position vector in the object, as described in post 22.14. When this result is spherically averaged it is independent of the directions of r and K and is given by
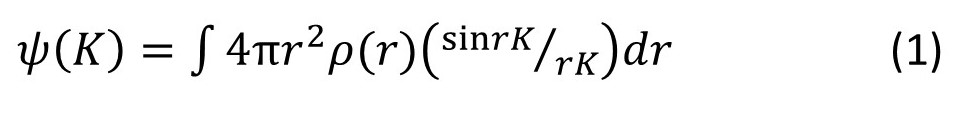
where r and K are the moduli of r and K, respectively. Equation 1 is derived in appendix 3; but you may need to read appendix 1 and appendix 2 first. When equation 1 is applied to an atom the integration is performed over all values of r for which ρ(r) is appreciable.
The value of sin(rk)/(rK) for K = 0, is 1 (see post 22.10), so that
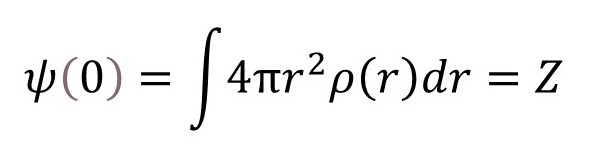
where Z is the atomic number – the number of electrons in the atom. So, for a hydrogen atom (Z = 1), ψ(K) has the shape shown in the graph below (see appendix 4 for more details).
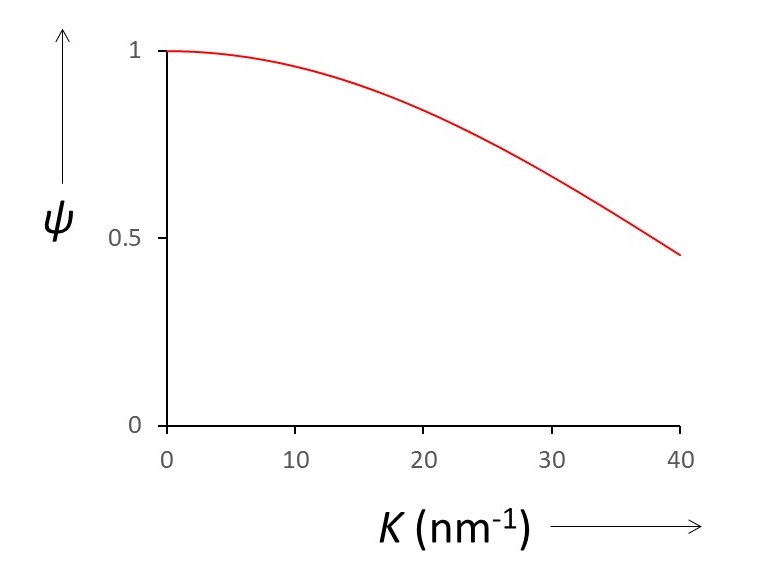
In most textbooks, ψ is represented by the symbol f and is called the atomic scattering factor for the atom. It is used whenever we want to calculate the diffraction pattern for an assembly of atoms in a molecule or in a crystal.
Related posts
22.4 X-ray diffraction
19.9 Electromagnetic waves
Follow-up posts
23.03 X-ray diffraction by a crystal
23.04 X-ray crystallography
25.8 The sphere
Appendix 1
The purpose of this appendix is to introduce spherical polar coordinates.
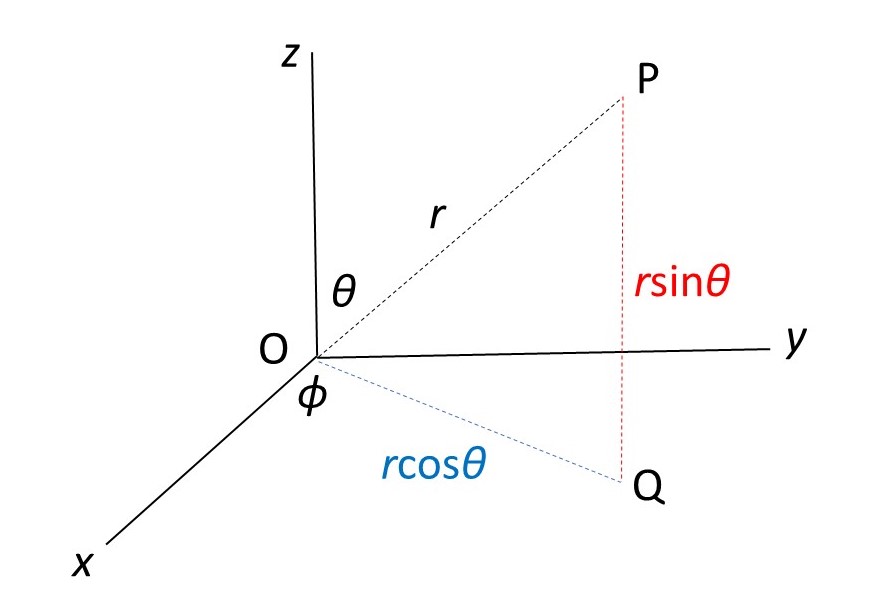
In the picture above, r is the distance of a point P from an origin O. OQ is the projection of OP on to the xy plane; φ is the angle between OQ and the x-axis, where 0 ≤ φ ≤ 2π (measuring angles in radians). The angle between OP and the z-axis is θ where 0 ≤ θ ≤ π. The range of θ values is less than the range of φ values because rotating θ by more than π radians is equivalent to simultaneous rotations of θ and φ.
The spherical polar coordinates of P are then r, θ and φ; its Cartesian coordinates are x, y and z.
From the picture above, and the definitions of sine and cosine (post 16.50), we can see that
x = rsinθcosφ
y = rsinθsinφ
z = rcosθ.
Since x, y and z can define any point in space, so can r, θ and φ. Note that this requires only two angles. So, to spherically average any function, we need average only about θ and φ. Averaging involves integration about these two angles. This is because, for a continuous distribution, adding up all possible values (to calculate an average, or a mean, is achieved by integration).
Appendix 2
Changing the mapping of a three-dimensional integral from Cartesian coordinates to spherical polar coordinates.
If you know about changing the mapping of an integral using a Jacobian operator (see post 22.16), this method will may seem very complicated – so you can ignore it. The explanation here is intended to use conceptually simpler methods.
If you don’t understand the first paragraph, let’s suppose that we want to integrate a function f(x, y, z) with respect to x, y and z – that is, we want to evaluate
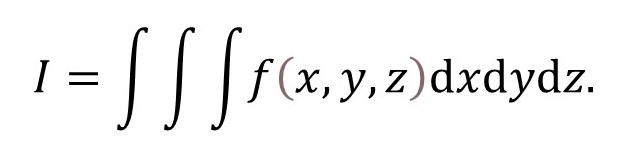
Now we transform f into spherical polar coordinates to get f(r, θ, φ). How do we calculate I?
In Cartesian coordinates an infinitesimally small volume in space is given by
δV = δx.δy.δz.
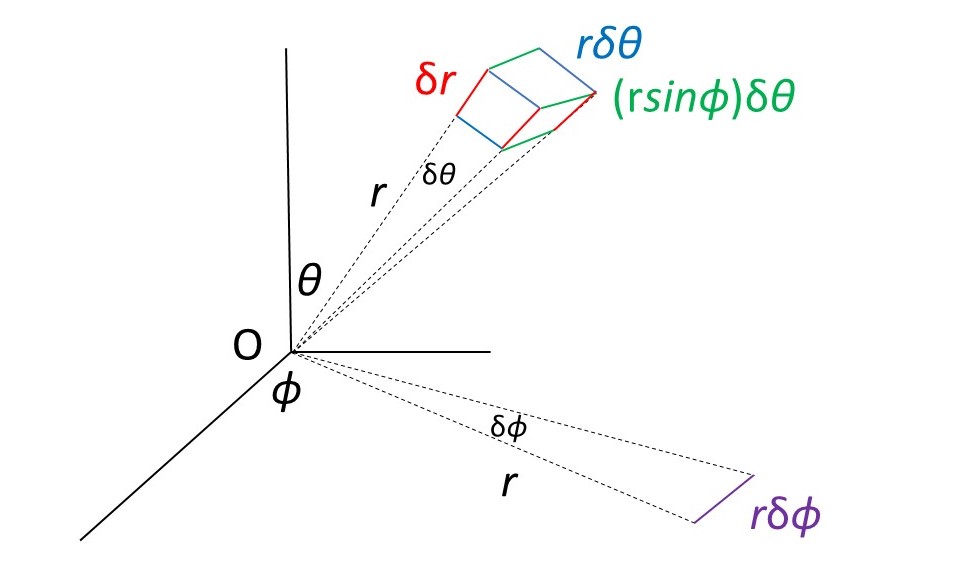
In the picture above, δV is obtained, in spherical polar coordinates, by multiplying the lengths of the infinitesimally short red, blue and green lines. To make the explanation simpler, think of the blue and green lines as arcs of a circle – if they are infinitesimally short, it makes no difference. The red lines subtend an angle δθ at the origin, O. The length of the red lines is δr. The length of the blue lines is rδθ (from the definition of a radian) The red, blue and green lines are at the end of an infinitessinally thin wedge that makes an angle θ with the x-axis. The purple line subtends an infinitesimally small angle, δφ, at O and makes the same angle, φ, with the x-axis as the infinitesimally thin wedge. The length of the purple line is rδφ, which (together with the two dashed lines passing through the origin) forms an infinitesimally thin triangle in the xy-plane. The wedge makes an angle θ with the z-axis. Since the z-axis is perpendicular to the xy-plane, the triangle makes an angle of (π/2 – θ) radians with the wedge. The green line is the projection of the purple line on to the wedge and so is equal to (rδφ)cos(π/2 – θ) = (rδφ)sinθ. Multiplying the lengths of the red, blue and green lines together gives the result that
δV = (δr).(δθ) (rδφ)sinθ = r2sinθ.δr.δθ.δφ
so that
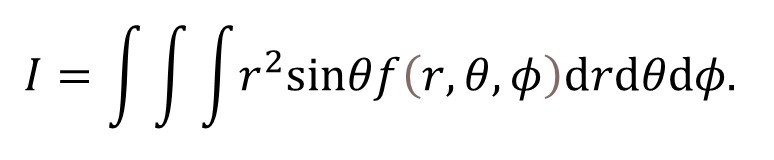
Many books state this result with very little explanation. The implication is that the result is obvious. As explained in post 17.1, we should beware an idea that obvious – it often means that someone doesn’t understand it or can’t explain it or, sometimes, that the idea is wrong.
Appendix 3
Derivation of equation 1.
Starting from
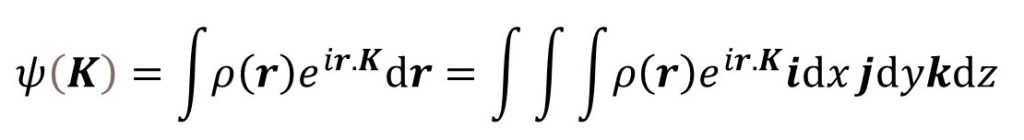
(where i, j and k are unit vectors in the direction of the x, y and z-axes, respectively) we make the final integral independent of the directions of the vectors – except that we need directions to calculate r.K. Then
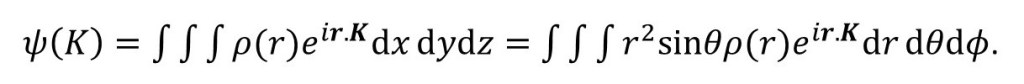
Let the angle between r and K be θ, then (from the definition of the dot product) r.K = rKcosθ.
Now let u = cosθ so that du/dθ = -sinθ.
Then we can write
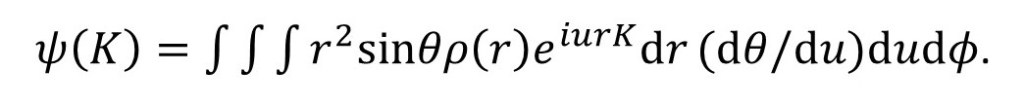
But sinθ(dθ/du) = sinθ/sinθ = 1, so that
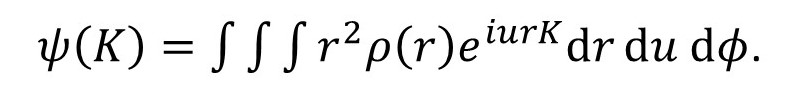
If we want to integrate with respect to φ over all possible orientations this integral becomes the definite integral.
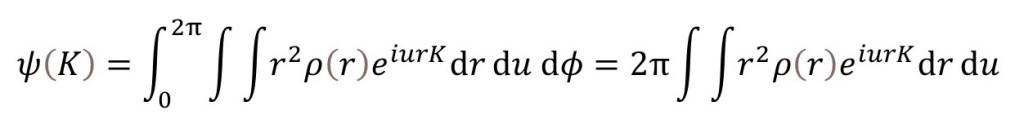
as explained in appendix 1. Since u is the cosine of an angle, when we spherically average with respect to u, the limits of integration must be -1 and +1 (the minimum and maximum values of the cosine of any angle). Then
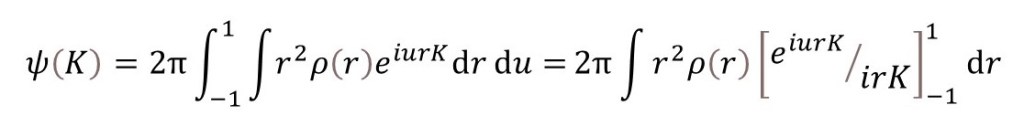
so that
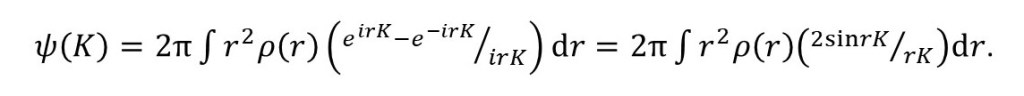
The final result comes from expressing sine as the difference between exponential functions and can be written as
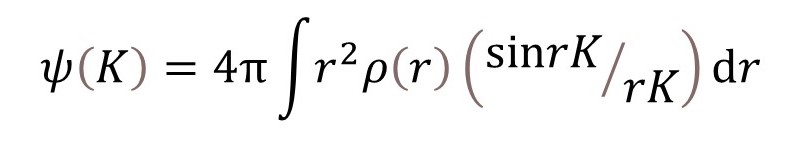
which is the required result.
Appendix 4
Atomic scattering factor for hydrogen.
The single electron in the hydrogen atom is in the 1s atomic orbital that has spherical symmetry. To calculate the atomic scattering factor, I should really use the wave function of this atomic orbital to find the probability of finding the electron at a given value of r (see post 19.28) and make the limits of integration between 0 and the value of r at which the probability of finding the electron is negligibly small. This is not easy. And all I want to do is indicate how the atomic scattering factor depends on K. I can do this by assuming that the electron is at the most probable value of r (called the Bohr radius). The is not really valid but does give an indication of the shape of the graph shown at the end of the main post. The Bohr radius for hydrogen is about 0.05 nm, allowing the approximate atomic scattering factor to be plotted against K.
For this simple calculation
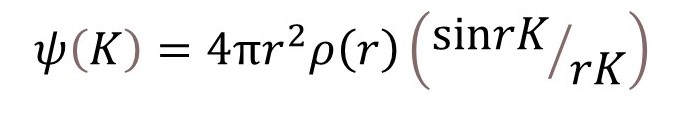
since the electron is at the Bohr radius. Remember that 4πr2 is the surface area of a sphere, so that ρ(r) is the atomic number, Z, that is 1 for hydrogen. If r = 0.05 nm, then

when K is measured in nm-1.