Before you read this, I suggest you read posts 22.14 and 22.21.

In post 22.21, we saw that an ideal crystal is an arrangement of atoms (that may be bonded together in molecules) or ions in a regularly repeating three-dimensional pattern. And, in post 22.14, we saw that the x-ray diffracted by an object could be represented, in both amplitude and phase by the Fourier transform of its electron density, ρ(r), given by
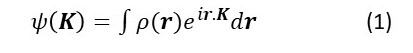
where K is the scattering vector whose modulus is given by
K = 4πsin(ϕ/2)(1/λ)
(see post 22.23); here ϕ is the angle through which the x-rays are scattered and λ is their wavelength. So to understand x-ray diffraction by a crystal, we need to be able to calculate the Fourier transform of its electron density.
We can consider an ideal crystal as the convolution of a lattice with the repeating arrangement of atoms or ions – called the unit cell (post 22.21). Therefore, the Fourier transform of the crystal is the Fourier transform of the electron density in a unit cell multiplied by the Fourier transform of the lattice. Since the electrons in the unit cell are associated with discrete atoms, the simplest way of thinking about the Fourier transform of the unit cell as the sum of waves scattered by the atoms in the unit cell – that is the sum of their structure factors. But, when we calculate this sum, we must remember the phase difference between the x-ray waves scattered by different atoms. In post, we saw that the phase associated with x-rays scattered by electrons whose position is given by the vector rj is exp(irj.K) where i is the square root of -1. Here exp(x) is just another way to write ex; I’ve written it in this form to make the text easier to read. The amplitude of the x-rays scattered by the jth atom in the unit cell is its atomic scattering factor, fj. So, when we add together the waves scattered by the atoms in the unit cell, the result is
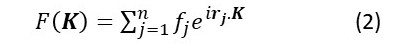
where the summation is over all the n atoms in the unit cell, and rj is now the position of the jth atom in the unit cell because the electrons are associated with discrete atoms.
Now we are going to multiply equation 3 by the Fourier transform of a lattice – to obtain the Fourier transform of a crystal. In post 22.24, we saw that the Fourier transform of a lattice was non-zero only at the reciprocal lattice points indexed by three integers, h, k and l – the Miller indices. Then, the values of K at which the Fourier transform of the lattice is non-zero were given by
K = ha* + kb* + lc* (3)
where a*, b* and c* are the reciprocal lattice vectors.
We can express rj as
rj = xja + yjb + zjc (4)
where xj, yj and zj are fractional unit cell coordinates of the jth atom in the unit cell and a, b and c are the unit cell vectors, as explained in post 22.21. From equations 2 and 4, the dot product of rj and K is given by
rj.K = (xja + yb + zc).( ha* + kb* + lc*) = 2π(hxj + kyj + lzj). (5)
The final step uses the Laue equations of post 22.24. So, when we multiply equation 2 by the Fourier transform of the lattice, the result is non-zero only when rj.K is given by equation 5. Therefore, we can write the Fourier transform of a three-dimensional crystal as
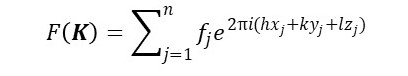
Since F(K) is non-zero only at the discrete reciprocal lattice points, we can write this result as
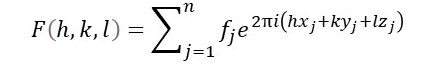
Now F(h, k, l) represents the x-rays scattered by the crystal, in both amplitude, and phase and is sometimes called its structure factor.
In post 22.14, we saw that
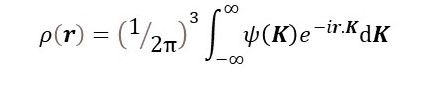
We normally ignore the scale factor (1/2π)3 because F(h, k, l) is not usually measured on an absolute scale. For an ideal crystal, we replace ψ(K) by F(h, k, l), that is non-zero only at integer values of h, k and l so that the integral above can be replaced by the Fourier series
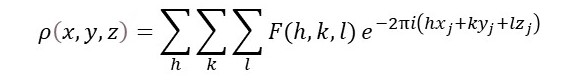
This is analogous to the derivation of the expression for F(h, k, l), above.
But to use this result to calculate ρ(x, y, z), the distribution of electrons (usually called the electron density) in a unit cell, we need to solve the phase problem, as explained in post 22.14.
Related posts
23.1 Observing x-ray diffraction by a crystal
22.24 Reciprocal lattice
22.23 K-space
22.22 Fourier transform of a one-dimensional lattice
22.21 An ideal crystal
22.14 X-ray diffraction
Follow-up posts
23.04 X-ray crystallography
23.10 Frequency analysis and x-ray crystallography