Before you read this, I suggest you read post 20.38.
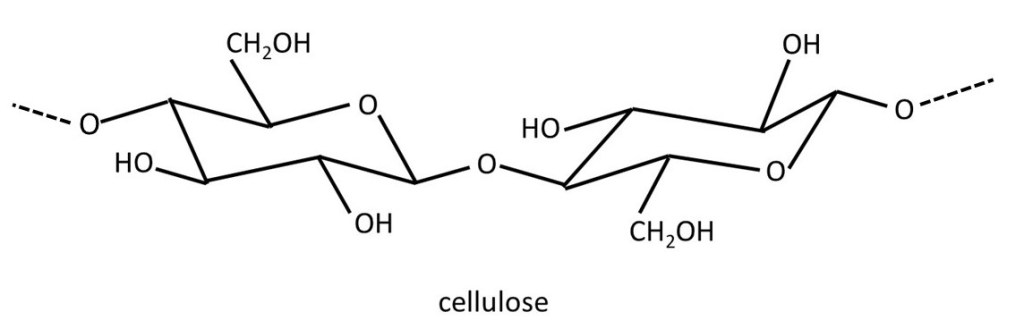
Cells (post 16.18) in plants are surrounded by a rigid wall made of the polysaccharide (post 20.38) cellulose that consists of repeating β-D-glucose residues (post 20.38) as shown above. In this picture, the second residue is identical to the first but I have shown it turned upside-down. The way I have drawn this structure, we can see the repeating 1 →4 linkages (post 20.38) joining β-D-glucose residues with their true shape. Hydrogen bonds (post 16.45) can form between adjacent cellulose molecules so that they form a rigid sheet. Post 20.38 explains the numbering scheme, as well as the meaning of α and β, used in describing linkages between the sugar residues in polysaccharides. But, if you wish you can ignore the description of these linkages when reading this post. Also, if you find the names of the sugar residues confusing, you can ignore them too.
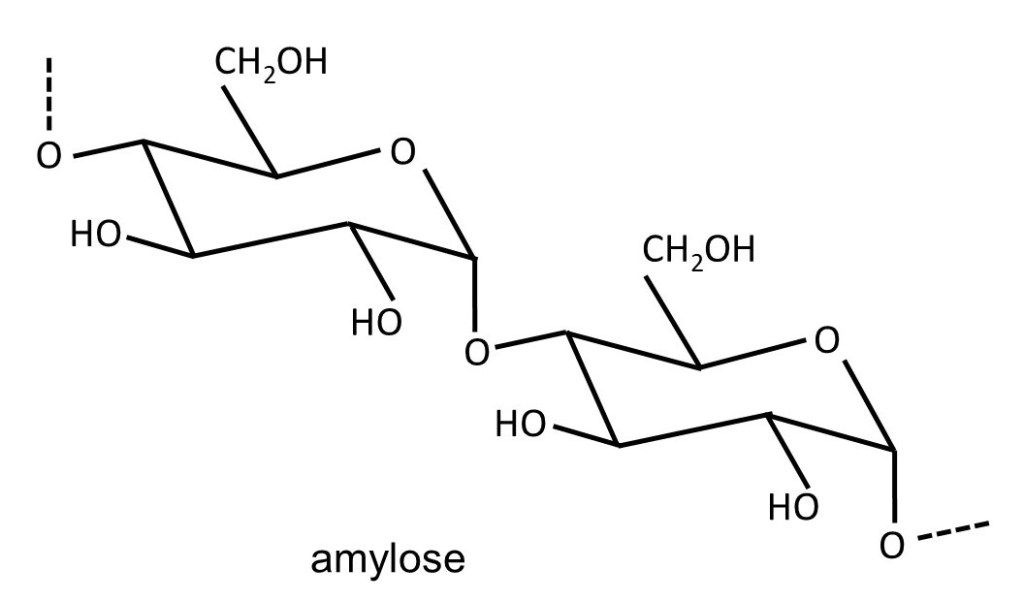
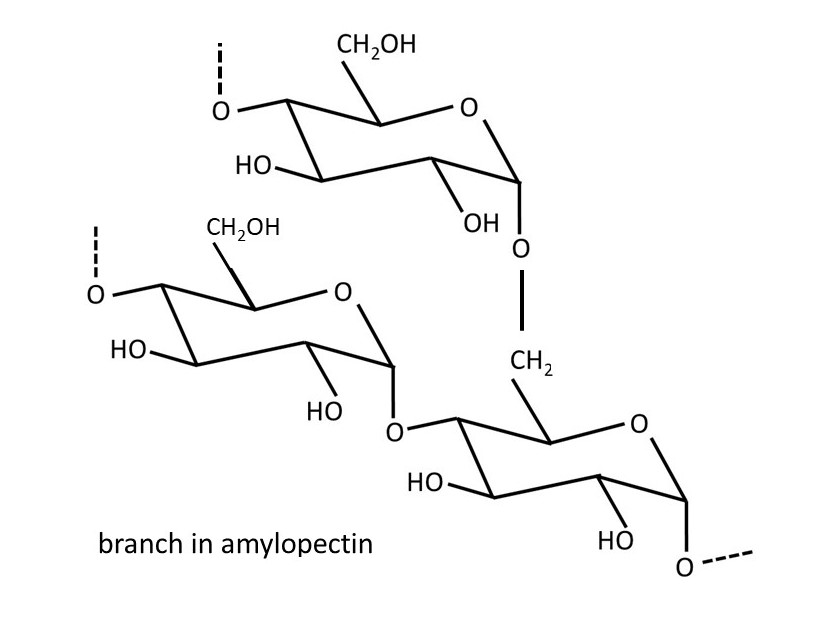
Plants make glucose from carbon dioxide and water in a process called photosynthesis; the activation energy for this reaction (post 16.33) is provided by sunlight absorbed by a green pigment called chlorophyll. This glucose is used to make adenosine triphosphate (ATP); hydrolysis of ATP provides the activation energy for the chemical reactions that keep the plant alive (post 16.47). If the plant makes excess glucose, it is stored as starch. Starch is a mixture of the polysaccharides amylose (shown above) and amylopectin. Amylose consists of α-D-glucose residues joined by 1 → 4 linkages. Amylopectin is almost identical but sometimes the polysaccharide chain branches because of the formation of 1 → 6 linkages, as shown below.
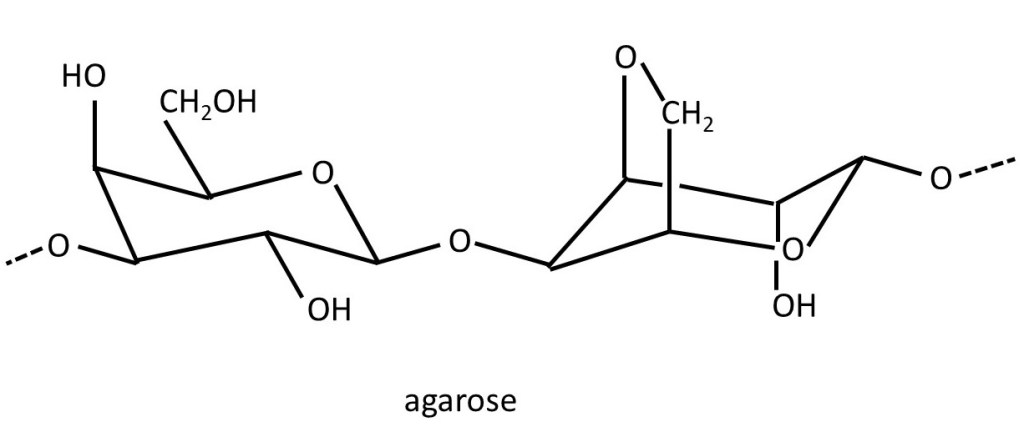
Agar, extracted from seaweeds, is sometimes used to form gels (post 20.39) in cooking and in the food industry. It is a mixture of the polysaccharide agarose and shorter polymer chains called agaropectins. The structure of agarose is shown below. It is a repeating disaccharide (post 20.38) consisting of an α-D-galactose residue (left-hand side) linked 1 → 4 to a 3,6 anhydro-L-galactose residue (left-hand side). These disaccharides are joined by 1 → linkages.
The structure of agarose shown above is a simplification because some -OH groups are replaced by a sulfate group. The structure of the sulfate group is often represented as -OSO3H, as shown on the left-hand side of the picture below. But this group occurs in the strong acid (post 17.49) sulfuric acid, and so it is almost completely ionised to form H+ ions and the -OSO3– ionic group (post 16.40), as shown below.
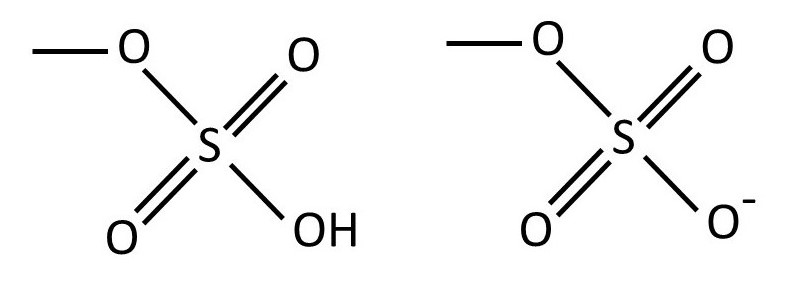
Calcium ions can bind to the ionised sulfate groups on different agarose chains to aid gel formation (post 20.39). Also, the -OH groups in the polymer can form hydrogen bonds with water molecules. Hence, agarose can form hydrogels (post 20.39).
Textbooks often concentrate on the polysaccharides found in plants. But polysaccharides are equally important for animals. For example, we make a form of starch called glycogen that is stored in the liver. Glycogen resembles amylopectin but has more branches.
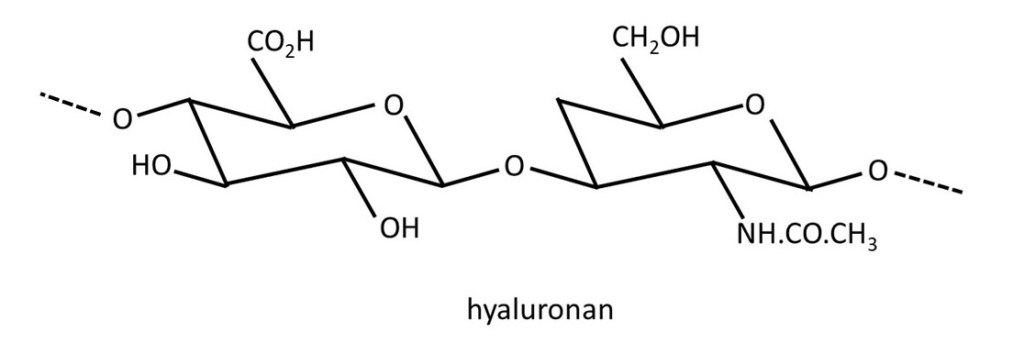
The picture above shows the structure of hyaluronan, sometimes called hyaluronic acid. It is a repeating disaccharide in which β-D-glucuronic acid is joined to β-D-N-acetylglucosamine by a 1 → 3 linkage. The disaccharide residues are then joined by 1 → 4 linkages. The structure of the -CO2H group is shown below (left-hand side). But, because of its similarity to acetic acid, it is a weak acid (post 17.49) and so exists as an equilibrium mixture with the -CO2– group whose structure is also shown below (right-hand side).
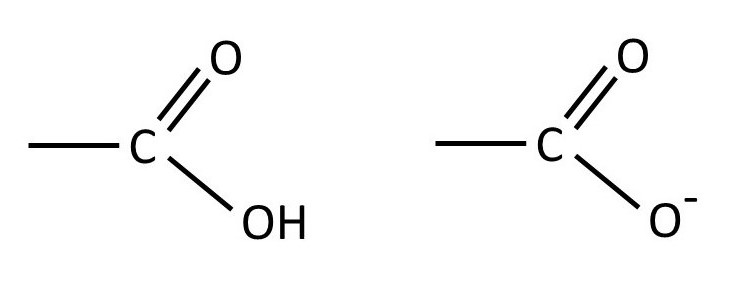
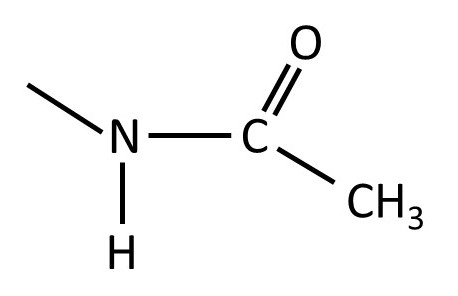
The structure of the -NH.CO.CH3 group is shown below.
Hyaluronan forms long polymer chains that aggregate shorter polymers in articular cartilage (posts 18.9 and 20.39) to form structures called aggrecans. These large structures prevent polysaccharides from being leached from the cartilage. The side groups on the hyaluronan molecule enable it to form hydrogen bonds (post 16.45) with water and so aid hydrogel formation (post 20.39). For example, it forms a gel in the interior of umbilical cord called Wharton’s jelly. It is used in surgery as a tissue filler. It also occurs in some cosmetic products for use on the face. I know of no evidence that it is absorbed by the skin and so I think it must simply fill wrinkles on the skin surface and not have any biological effect. So, it seems to me, that these products are really not much different to any other facial foundation creams.
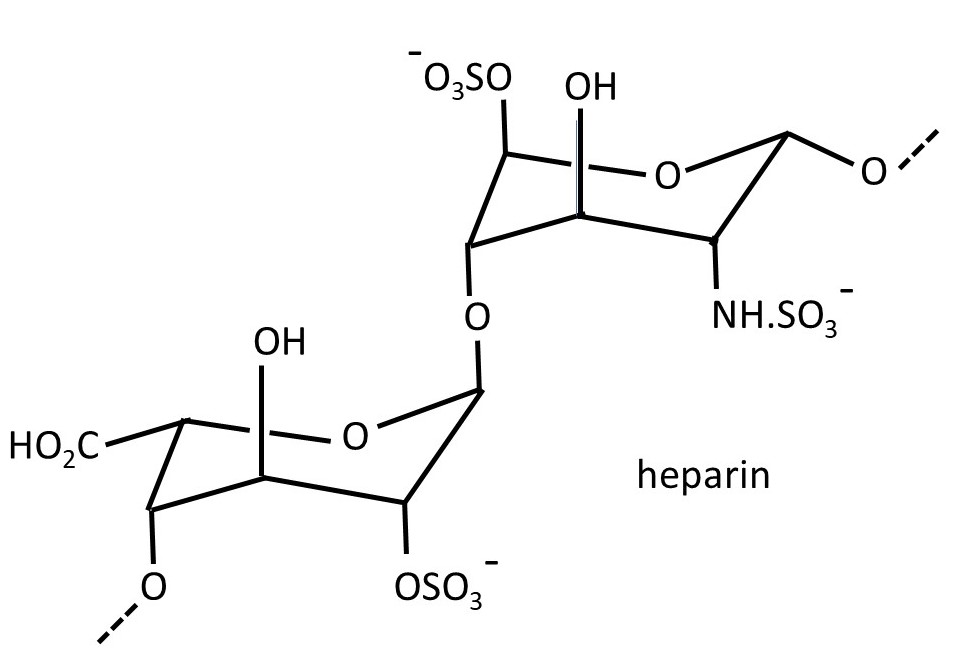
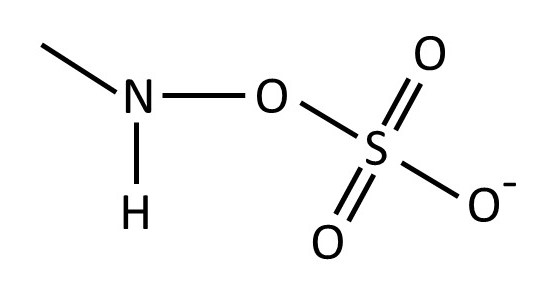
The picture above shows the structure of heparin – a polysaccharide secreted by specialist cells (post 16.18) in our bodies called mast cells. It is a repeating disaccharide consisting of 2-O-sulfated iduronic acid residues (left-hand side) joined by α(1 → 4) linkages (post 20.38) to 6-O-sulfated, N-sulfated glucosamine residues; the disaccharide residues are joined by β(1 → 4) linkages. The structure of the -NH.SO3– group is shown below.
However, this is a simplified version of the structure of heparin because the sulfate groups don’t always appear in the same place. Heparin is an anticoagulant (it inhibits blood clot formation) and is involved in the complicated series of reactions involved in the control of blood clot formation. It is often injected after surgery to prevent unwanted blood clots from forming in veins.
These are a few examples to show the importance of polysaccharides in biology, food production and medicine.
Related posts
20.39 Gels
20.38 Sugars
20.7 Polymers
16.47 Fats
16.30 Molecules
Follow-up posts