Before you read this, I suggest you read post 20.32.
In the English language the word “sugar” is often used to mean “sucrose” – the sugar extracted from sugar cane or sugar beet that is often used to sweeten foods and drinks. But sucrose is not the only type of sugar – other examples are glucose, fructose and lactose.
How can we define a sugar? I have never found a good definition. Sugars are usually defined as sweet-tasting carbohydrates that are soluble in water. Carbohydrate molecules contain only carbon, hydrogen and oxygen atoms with usually (but not always) twice as many hydrogen atoms as oxygen atoms (see post 16.30 to find out more about molecules). The phrase “but not always” makes it clear that this is an unsatisfactory definition. The definition is even worse than it might appear. This is because acetic acid, C2H4O2 (post 17.49), fits the definition but isn’t a carbohydrate! So sugars are defined by something that can’t be defined. I also don’t like the words “sweet-tasting” in the definition of sugars. This is because taste is subjective – it is possible that something that tastes sweet to most people won’t taste sweet to everyone. This is unsatisfactory because science is intended to be objective and not subjective (posts 16.22 and 16.36). A further problem is that it is not recommended that we taste unknown substances – they may be poisonous. Suppose someone discovers a new substance that could be a sugar. If he or she tastes it, it could be so poisonous that they are killed before the result can be reported! So, how do we know whether it is sweet?
How can I write about something that I can’t define? I can’t define the word “dog” but I have seen so many dogs that I know what one looks like; so I am confident that I know whether an animal is a dog. In this post, we are going to see what molecules of sugars look like. Then, perhaps, will have a better idea of what a sugar is.
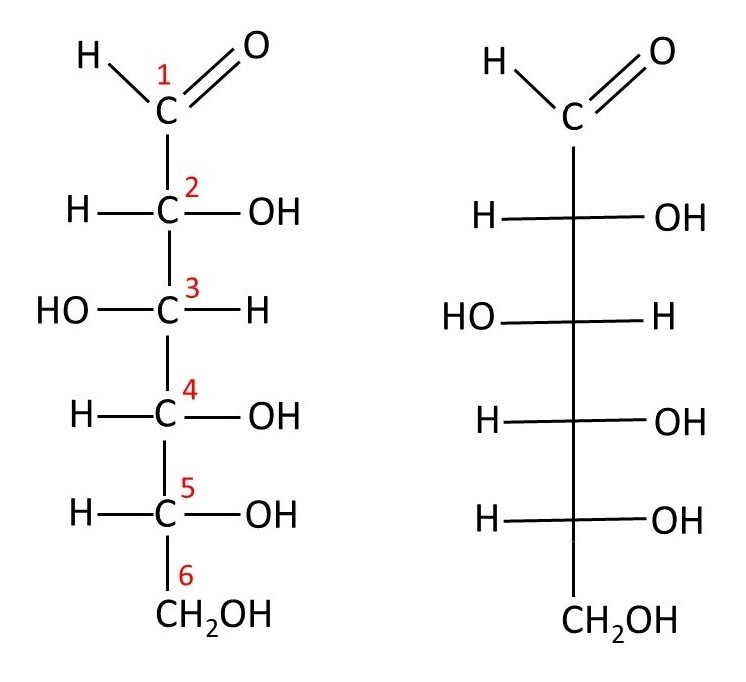
Although sucrose is the sugar that most people know best, I am going to start by writing about glucose because it is a simpler example of a sugar. Glucose (C6H12O6) has the structural formula shown above. On the left-hand picture the numbers in red are the numbers that are conventionally used to label the six carbon atoms. This left-hand picture tells us nothing about the shape of the molecule. But we could consider this picture to be a Fischer projection of the shape of the molecule. Then the horizonal bonds from carbon C2 point towards us. Remember that the four single bonds attached to a carbon atom point towards the corners of a tetrahedron (post 16.30). Now we can, with some difficulty determine the three-dimensional shape of the molecule. Note that it is chiral (post 20.27); the molecule shown here is the optical isomer called D-glucose – this is the form of glucose that occurs naturally. Because it rotates the plane of plane polarised light to the right, it can also be called D(+)-glucose (post 20.29). To show that our structural formula is a Fischer projection, it is common to write it like the right-hand side of the picture above; in this formula, there is a carbon atom wherever two bonds cross.
But the formula above doesn’t show us the true shape of a D-glucose molecule because, when dissolved in water, the oxygen atom on C1 can react (post 16.33) with the -OH group on C5 to form the a ring of six atoms, as shown below. Compounds whose molecules form rings are called cyclic compounds. The left-hand side shows all the atoms, except for the carbon atoms where the bonds in the ring cross. It is usual to miss out the hydrogen atoms that are bonded directly the carbon atoms (as in the right-hand side picture below); then, if a carbon atom appears to form less than four bonds, the missing bonds are bonds to a hydrogen atom.
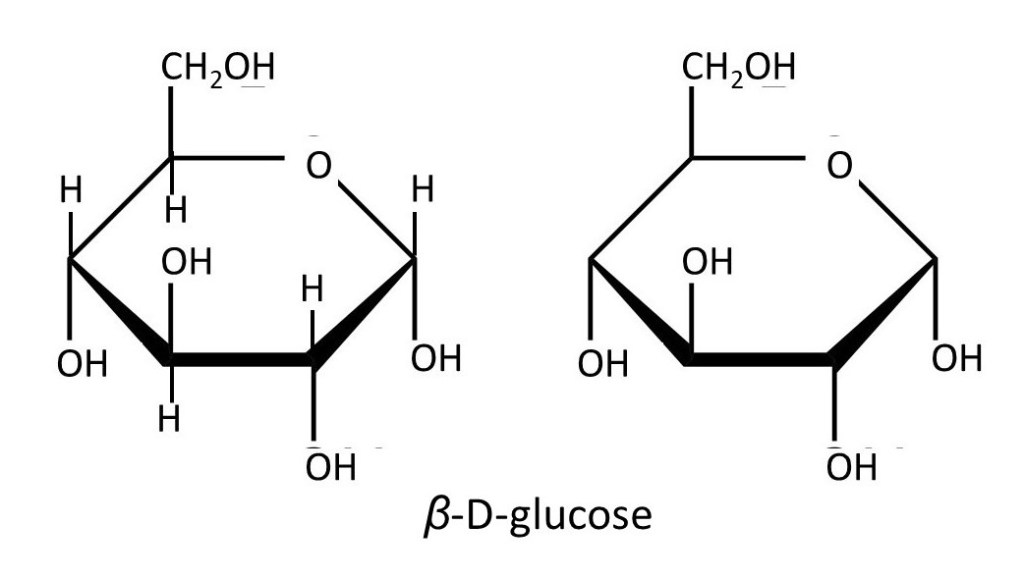
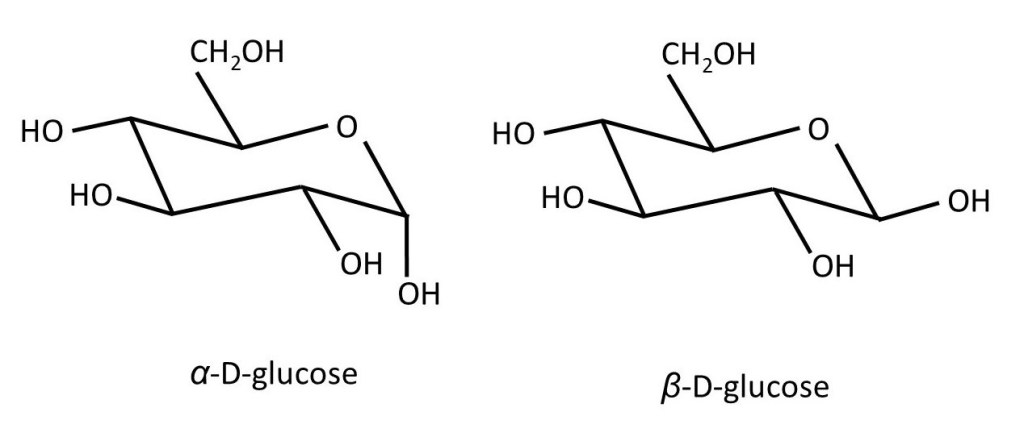
The formulae shown above can be very useful – but they still don’t show us the true shape of the molecule. The six-membered ring isn’t flat – it can’t be because the single bonds from the carbon atoms point towards the corners of a tetrahedron (post 16.30). So, the pictures below show us the true shape of the D-glucose molecule. Note that, when the ring is formed, the -OH group on C1 can point either sideways (the equatorial direction) or downwards (upwards and downwards are called the axial direction). The first form is called β-D-glucose; the second is called α-D-glucose. In a sample of D-glucose, most of a molecules are in the β form and some are in the α form; the two exist in equilibrium.
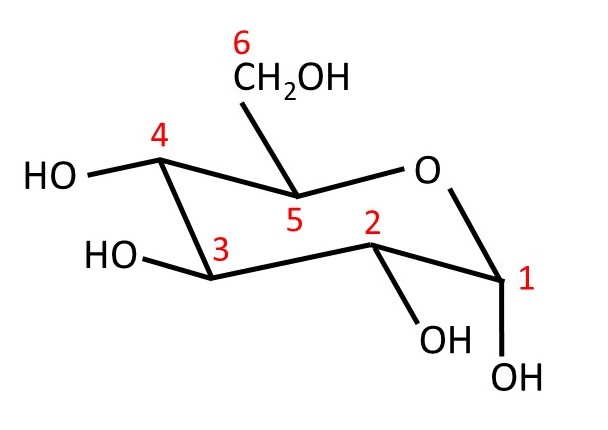
α and β-D-glucose are two structural isomers (post 20.32) that can change from one to the other so easily that they exist in equilibrium. Structural isomers that behave in this way are called tautomers and this behaviour is called tautomerism. The two tautomers of D-glucose (and other sugars), where the -OH group at C1 can be either axial or equatorial, are sometimes called anomers. At this point, it might be useful to look at the numbering scheme for the carbon atoms in these cyclic forms of D-glucose – shown in the picture below.
Since the D-glucose molecule is not identical to its mirror image, there are two more possible forms of glucose shown in the picture below – see posts 20.27, 20.29 and 20.32, for more details on molecules that are not identical to their mirror images.
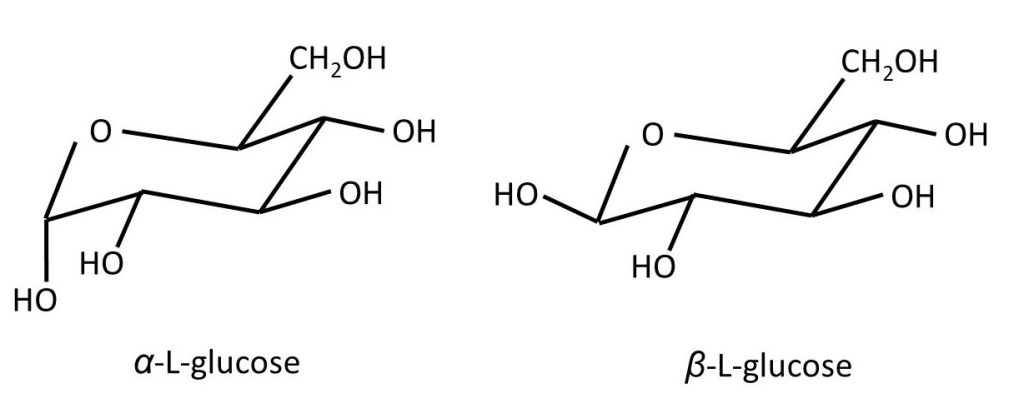
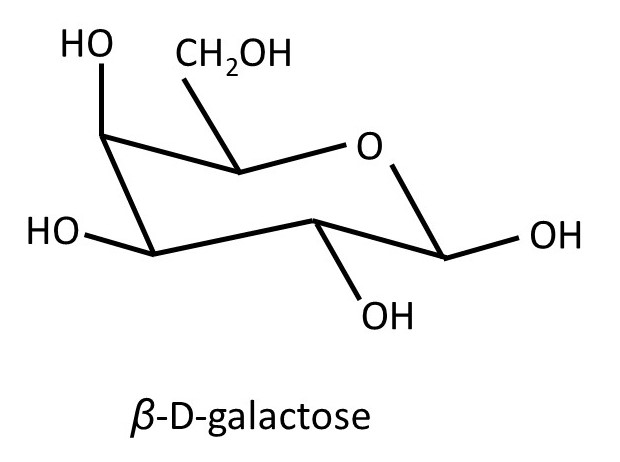
So; when the position of the -OH group on C1 changes from axial to equatorial, the molecule is still glucose; but if the configuration at C2, C3, C4 or C5 changes, a different compound is formed. Here configuration refers to different shapes of molecules that can only be converted from one to the other by breaking a covalent bond (post 16.30). For example, the picture below shows the structure of β-D-galactose.
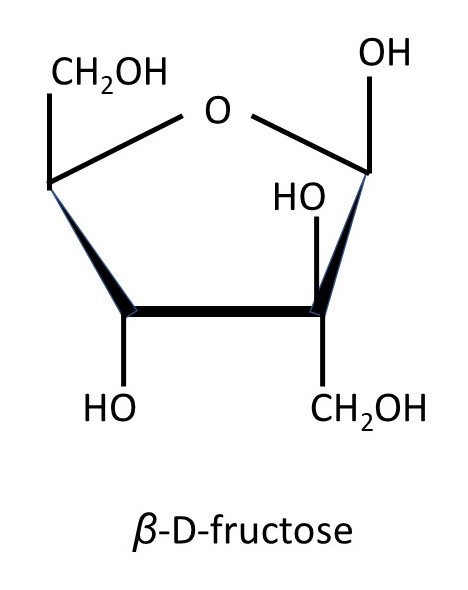
The sugars that can be formed in this way contains six carbon atoms and are called hexoses. If they can form rings of six atoms, like glucose and galactose, they can also be called aldohexoses. Not all hexoses can form rings of six atoms. D-fructose, the sugar that occurs naturally in fruit, is a hexose that forms a ring of five atoms – as shown in the picture below.
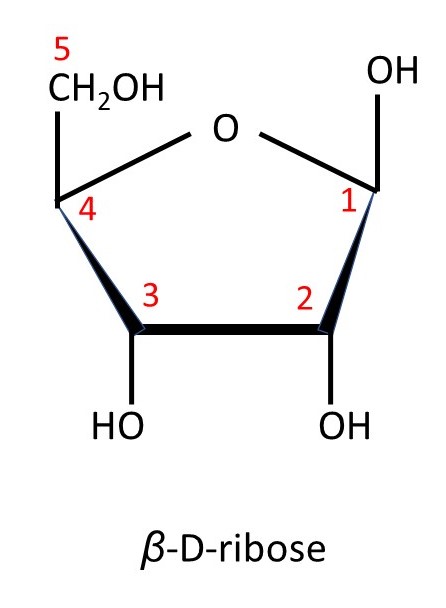
Some sugars have molecules that contain five carbon atoms – they are called pentoses. The picture below shows an example – D-ribose. The conventional numbering scheme for the carbon atoms in a pentose are shown in red.
So far, we have looked at sugars that have only one ring; they are called monosaccharides. But two sugar rings can covalently bond (post 16.30) to form a disaccharide. Sucrose (see the first paragraph) and lactose (the sugar in milk) are examples of disaccharides.
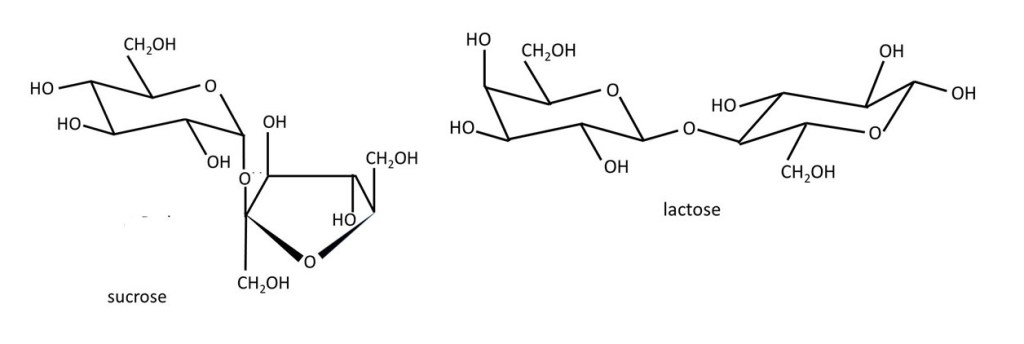
In sucrose, an α-D-glucose molecule (left-hand side) is bonded to a β-D-fructose molecule. If this isn’t clear, notice that I’ve turned the β-D-fructose molecule, shown previously, upside-down. Note that the glucose and fructose molecules have lost an oxygen atom and two hydrogen atoms to form sucrose. So we say that sucrose consists of an α-D-glucose residue and a β-D-fructose residue. The use of the word “residue” here is the same as in the description of condensation polymers (post 20.7). The bond between these residues is from C1 (glucose) to C2 (fructose); so we call this a 1 → 2 linkage. In lactose, a β-D-galactose residue makes a 1 → 2 to a β-D-glucose residue. Note that, this time, I’ve turned the β-D-glucose residue upside-down (when compared to the previous picture of this molecule) to make the structure clear.
Oligosaccharides have between three and ten sugar rings bonded together. Polysaccharides are polymers (post 20.7) made of sugar rings – they will be the subject of a later post.
I’m sure you can recognise a dog when you see one – now I hope you will be able to recognise a sugar molecule!
Related posts
16.30 Molecules
16.47 Fats
20.32 Isomerism
Follow-up posts