Before you read this, I suggest you read posts 16.21 and 16.30.

A chemical reaction occurs when atoms change the way in which they share electrons to form new molecules (see post 16.30). For example, a carbon atom (C) reacts with an oxygen molecule (O2) to form a molecule of carbon dioxide (O=C=O or CO2). When something reacts with oxygen in this way, we say that it “burns”. We can represent this reaction by the chemical equation:
C + O2 → CO2.
This isn’t really an equation like x = 3, because an equations has an “equals” (=) sign that, in this example, means that what is on its right-hand side (3) is identical to what is on its left-hand side (x). The chemical equation is different; it shows that a carbon atom can react with an oxygen molecule to make a carbon dioxide molecule. The only sense in which it is an equation is that the number of atoms on the right-hand side (1 carbon and 2 oxygen) must be the same as on the left-hand side because all that happens in a chemical reaction is that the same atoms share their electrons in a different way.
Another example is that nitrogen molecules (N2) can react with hydrogen molecules (H2), under the right conditions, to form ammonia molecules (NH3). This reaction is used in the Haber-Bosch process for producing ammonia from atmospheric nitrogen to make artificial fertilizers; some plants (peas and beans) make atmospheric nitrogen into plant food naturally. The chemical equation that represents the reaction between nitrogen and hydrogen is NOT:
N2 + H2 → NH3.
The reason is that the left-hand side Has 2 nitrogen atoms and 2 hydrogen atoms; the right-hand side has 1 nitrogen atom and 3 hydrogen atoms. However, we can correct this problem by writing the equation as:
N2 + 3H2 → 2NH3.
This means that one nitrogen molecule reacts with two hydrogen molecules to make two ammonia molecules.
How much carbon dioxide do we make when we burn 1.0 kg of carbon? Carbon has a relative atomic mass of 12 (see post 16.27). A carbon dioxide molecule contains 1 carbon atom (relative atomic mass 12) and 2 oxygen atoms (each with relative atomic mass 16); so the relative molecular mass of carbon dioxide is 12 + (2 × 16) = 12 + 32 = 44. So the mass of carbon dioxide produced by burning 1.0 kg of carbon is (1.0/12) × 44 = 3.7 kg. Charcoal is an impure form of carbon so, when you burn 1.0 kg of charcoal on a barbecue, you make about 3.7 kg of carbon dioxide.
Why do chemical reactions occur? The reason is that the products of the reaction (on the right-hand side of the equation) must be more stable than the reactants (on the left-hand side of the equation). Being “more stable” means having less energy, in exactly the same way as a ball rolls downhill to minimize its potential energy (see post 16.21). This energy is given out, usually in the form of heat – so we use the energy given out when charcoal burns to cook food. However, energy can be given out as sound (in an explosion) or by creating an electrical current. Reactions that produce electric currents involve ions (the subject of a later post) and occur in batteries. In our muscles, the energy released by the chemical reaction when an adenosine triphosphate (ATP) molecule is converted into adenosine diphosphate (ADP) is used to directly produce mechanical energy.
But we usually have to add some energy to get a reaction started – in the same way that we may need to give a ball a push, to overcome friction (see post 16.19), before it can roll downhill. The energy needed to start a chemical reaction is called the activation energy. The reason we need to add this energy is that the reactants are stable (low energy) and have to move their electrons into less stable (higher energy) levels, to form a transition state, before more stable bonds can be formed to produce more stable (lower energy) products. For example, we have to add some heat energy to charcoal to make it burn – it then releases more heat.
The energy levels for a reaction are shown in the diagram below.
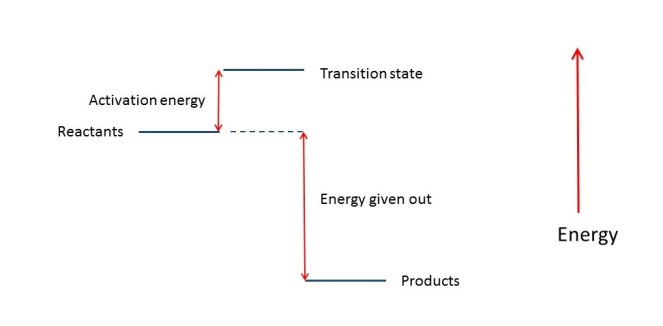
Sometimes, we can lower the energy of the transition state, and so lower the activation energy, by adding a substance called a catalyst. A catalyst is something that can form temporary bonds with the transition state and so make it more stable. Lowering the activation energy makes it easier for reactants to form the transition state and so form products – as a result, the catalyst makes the reaction occur more quickly. The Haber-Bosch process (see above) uses powdered iron as a catalyst.
Chemical reactions are important because they enable us to make new materials that are useful to us – for example, to make medicines, to convert latex into rubber suitable for making car tyres (vulcanization), to make materials (like polyesters and nylon) that can be used to make clothes and very many more applications. They can also be used to produce heat (for cooking, to heat our homes, to convert into mechanical energy – for example in car engines) and to produce electricity (either directly in batteries or indirectly by converting the heat they produce into electricity).
Related posts
16.31 Electrons in molecules
16.30 Molecules
16.29 Electrons in atoms
16.27 Atoms
16.21 Energy
Follow-up posts
19.5 Oxidation and reduction
19.6 Cells and batteries
19.7 Redox potentials
22.17 Kinetic stability