
The picture above shows how carbon, hydrogen and oxygen molecules combine to form a fat molecule; as expected each carbon atom forms 4 covalent bonds, each hydrogen atom forms 1 covalent bond and each oxygen atom forms 2 covalent bonds. (More information on covalent bonds and molecules is given in post 16.30.) The vertical bonds in this picture appear longer than the horizontal bonds – this is to make it easier to draw the picture; really the bonds are all approximately the same length.
R’, R’’ and R’’’ represent chains of covalently bonded carbon atoms with hydrogen atoms bonded to them, like C17H35– in post 16.40.

The picture above shows a short chain of this kind: C6H13-. This can also be represented as CH3.CH2.CH2.CH2.CH2.CH2-. The carbon atom bonded to three hydrogen atoms is sometimes called the ω (Greek letter “omega”) carbon atom. It can also be labelled as carbon atom number 1; the others are labelled 2, 3, 4…. as shown above.
If we continue the chain in the picture, we get CH3.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2.CH2– which is the same as C17H35-.

In C17H35– all the carbon atoms in the chain (apart from the ω carbon atom) have two hydrogen atoms bonded to them, as shown in the picture above (left-hand side). Fats that contain chains like this are called saturated fats. In some fats, some of the carbon atoms are joined to each other by two bonds (picture above, right-hand side). The carbon atoms that are joined by two bonds are bonded to only one hydrogen atom. Notice that all the carbon atoms still form 4 bonds and all the hydrogen atoms still form 1 bond, as expected. Fats that contain chains with these carbon-carbon double bonds are called unsaturated fats. Fats that contain chains with several of these double-bonds (typically about 4) are called polyunsaturated fats.

Some unsaturated fats are trans-unsaturated, as shown in the picture above (left-hand side), others are cis-unsaturated (right-hand side). The two are different because rotation around the C=C double bond is not allowed, although rotation can occur around the C-C single bond. The bonds shown in the picture above all lie in a plane and make angles of about 120o with each other. Reasons for these statements are given, very briefly, in the appendix, at the end of this post; most people will want to ignore it!
Fats are an important part of our diet. However, there is a lot of confusion about this. One reason for the confusion is that saturated fats tend to be solids and unsaturated fats tend to be liquids. Liquid fats are often called oils. So olive oil and fish oils are fats.

When we digest fats, we break them down to make fatty acids; a fatty acid molecule is shown in the picture above. We can convert fatty acids into stuff called adenosine triphosphate (ATP). The energy (post 16.21) released when ATP is broken down into adenosine diphosphate (ADP) and phosphate ions is required for most of the functions of our bodies. However, we can also make ATP from carbohydrates.
If the lowest number carbon atom involved in a double bond is number 3, and there are further double bonds along the carbon chain, then we have an omega-3 polyunsaturated fat. An example of an omega-6 polyunsaturated fatty acid is:
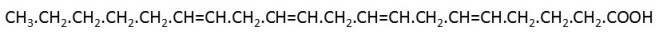
The fatty acids alpha-linolenic acid (an omega-3 polyunsaturated fatty acid) and linolenic acid (an omega-6 polyunsaturated fatty acid) are essential for health. They are called essential fatty acids and are needed by the brain and nervous system. We obtain these essential fatty acids from nuts and seeds, eggs and oily fish.
We store excess fatty acids as fats. Excessive fat storage may be a problem but we need to store some as a long-term store of fatty acids for providing energy.
There are many opinions about adverse effects of fat consumption. Many nutrition “experts” seem to base their opinions on outdated or non-existent scientific evidence or don’t appear to understand the need for evidence to support their beliefs. There are very many articles, books and webpages on the subject that are completely misleading.
I have searched the scientific literature to find out what is currently known about the adverse effects of fat consumption. This literature is made up of the journals in which scientists publish their original research. All articles are reviewed to ensure that the studies were sensibly conducted and that the results obtained justify the conclusions. This reviewing process is not perfect but it is better than nothing. More importantly, we can read these articles and think about the evidence for ourselves – instead of simply accepting that whatever we read must be true! This is much better than reading articles in everyday magazines where all the evidence is not fully available.
I was lucky! In 2015 the British Medical Journal published an article that combined the results from many reliable studies. It is available at https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4532752/. If you’ve never seen any original scientific research reports, it’s worth looking at this and comparing it with the magazine articles that you’ve read on the same subject. The authors could find no evidence that saturated fatty acids were associated with any health problems – including cardiovascular disease, coronary heart disease, stroke or type 2 diabetes; however, they could not be as confident as they would like to be about this conclusion. Trans-unsaturated fatty acids were associated with all these problems. Remember that this association does not show that trans-unsaturated fatty acids cause these problems (see post 16.10) but they may do.
Some trans-fatty acids occur in the milk and body fats of animals that have four stomach compartments so that they can digest cellulose – including cattle, sheep, goats and deer. Interestingly, this list of animals does not include horses or rabbits. However, for most people, the main source of trans-unsaturated fats is in industrially-produced food, because they are cheaper than animal fats and have longer shelf-lives. They are made by chemical modification of naturally-occurring fats. Food that contains trans-unsaturated fats may not be obviously “fatty” – they can occur in all sorts of bread and biscuits (“cookies” in the USA) and snack foods.
But remember – when we evaluate all the information about fats and health, it is important to remember that the conclusions of any scientific research may have to be modified as further evidence becomes available (see posts 16.2 and 16.3).
Related posts
16.45 Water
16.36 Good and bad
Follow-up posts
16.48 How does soap work?
20.38 Sugars
25.2 Sigma bonds and pi bonds (explanation of why rotation about C=C double bonds not allowed)
Appendix
The two p atomic orbitals that form the second bond can only overlap if the bonds shown in this picture lie in a plane – post 16.30; alternatively, we can think of the second bond as being the same as a π molecular orbital in which the LCAO approximation only works if the bonds are in the same plane, because of the symmetry of the p orbitals that we need to combine – post 16.31.