Before you read this, I suggest you read posts 16.30 and 16.33.
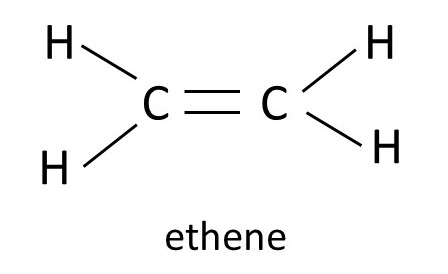
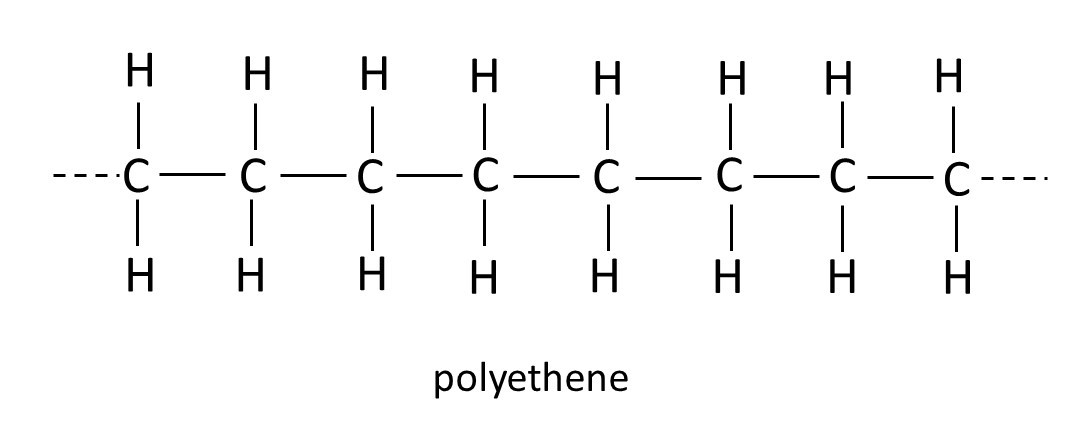
At high pressure, in the presence of a suitable catalyst (post 16.33) ethene molecules (see picture above) join to form polyethene or polyethylene or polythene, whose formula is shown below. Notice that each carbon atom is still bonded to two hydrogen atoms, as in ethene, but is joined to two carbon atoms (one from the original ethene molecule and the other from another ethene molecule that takes part in the reaction) by single bonds. This chemical reaction (post 16.33) is called the Ziegler-Natta process and is used for making polyethene on an industrial scale. A molecule, like polyethene, that is made by joining smaller molecules is called a polymer. And each small molecule that is used to make the polymer is called a monomer.
This formula of polyethene, shown above, does not represent the shape of the molecule. Remember that four single bonds formed by a carbon atom point towards the corners of a tetrahedron (post 16.30). So the shape of the polyethene molecule is shown in the picture below (where a black ball represents a carbon atom and a white ball a hydrogen atom).

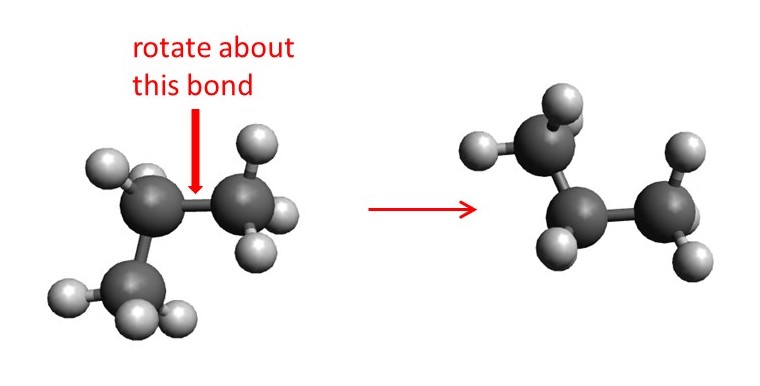
This picture shows a fully extended polyethene chain. Now let’s think about any of the C-C single bonds. Rotation about this bond changes the shape of the molecule, as shown below.
When rotations like this occur along the length of the chain, the molecule can fold in three dimensions: an example is shown below.
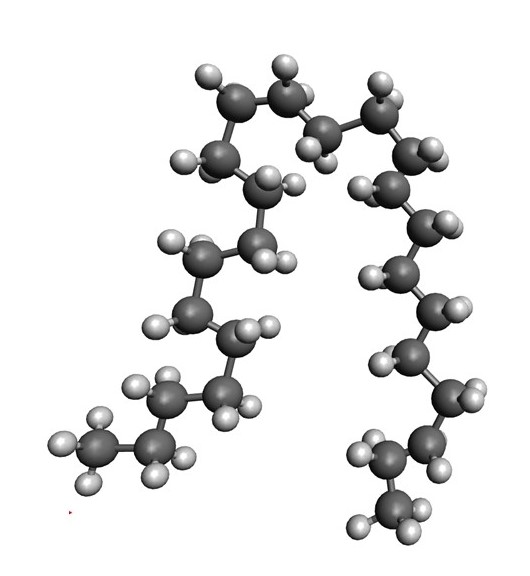
When polyethene is made, many of the chains will fold and tangle. As a result, it is difficult for an individual polyethene molecule to move. So, although ethene is a gas, at atmospheric pressure and normal temperatures, polyethene is a solid (see post 16.37).
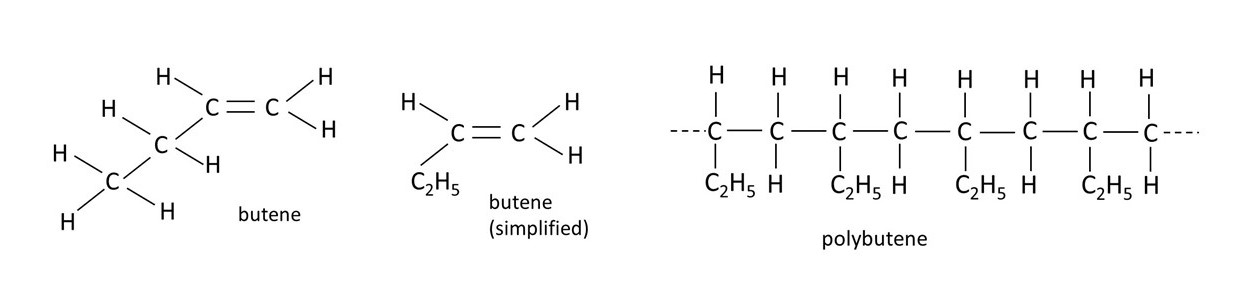
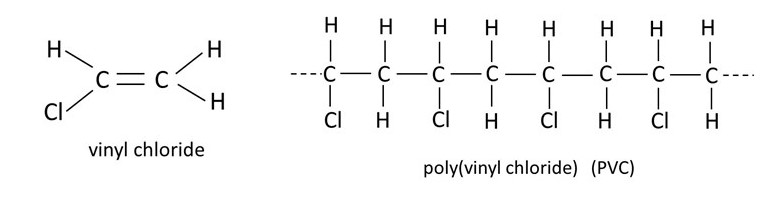

Some other examples of monomers, like ethene, and the corresponding polymers, are shown in the picture above. They all have familiar everyday applications; polybutene (polybutylene) is used to make water pipes, PVC can be used to make clothing and polystyrene is commonly used in packaging.
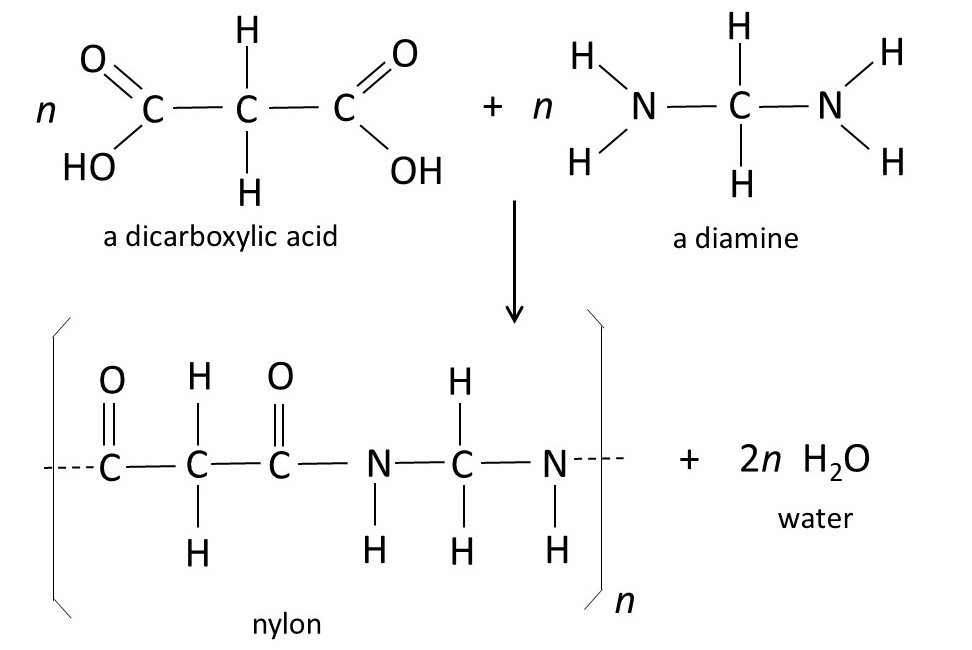
The picture above shows the formulae of a dicarboxylic acid (so called because it has two carboxylic acid groups, -COOH, like the acidic groups in acetic acid, post 17.49) and a diamine (so called because it contains two -NH2, or amine, groups). A chemical reaction (post 16.33) can join one of these molecules to another, with the loss of a water molecule (H2O, post 16.30), to make one type of nylon. In this example, polymerisation isn’t simply about joining monomers – other, smaller molecules are lost in the process; in this example the smaller molecules are water. When polymers form with the loss of smaller molecules, the process is called condensation polymerisation and the result is a condensation polymer. The repeating unit in the polymer is sometimes called a monomer residue, because it does not contain all the monomer atoms.
Not all samples of the same polymer are the same. For example, polyethene can be used to make flexible bags and to make a rigid component of artificial hip joints. The higher the relative molecular mass (post 16.33) of the polymer, the more tangled the chains become. Then they are very difficult to pull apart and the solid polymer is very stiff. The form of polyethene used to make artificial hips is called “ultra-high molecular weight polyethylene” – it has a very high relative molecular mass. (Relative molecular mass is sometimes called “molecular weight” – see post 16.17 to find why this is an incorrect use of the word “weight”). It is, therefore, very stiff and can form a rigid component of a joint. Polyethene food bags are made a polyethene with a lower relative molecular mass and so are more flexible. Also, polyethene for hip joints is made in blocks that can be cut into three-dimensional shapes, using the same techniques as would be used for a soft metal. Polyethene for food bags is rolled into thin sheets – because these sheets are thin, they are flexible (post 20.2). Nylon can be manufactured as a block, for some applications, or pulled into a thread for weaving into clothes. PVC is rolled into sheets to make a shiny fabric used in clothes.
In future posts I hope to write about many more types of polymers and why they are important materials for making things, as foods and in biological processes. There are very many applications of materials based on polymers. These materials are usually called “plastics”. Unfortunately, many polymers are very difficult to recycle. As a result, polymer waste is becoming a serious environmental problem – an example where applying science brings problems as well as benefits (see post 18.18).
Related posts
16.37 Solids, liquids and gases
16.30 Molecules
Follow-up posts