Before you read this, I suggest you read posts 16.30, 16.33, 16.39 and 17.48.

In 2017 there was an alarming increase in attacks in London in which victims had acid thrown in their faces. One way in which these attacks could be reduced might be to make it illegal for people to carry acids in public places.
However, it appears that any new laws will refer not to “acids” but to “corrosive liquids”. Why? There are two reasons. One is that not all corrosive liquids that could be used to attack people are acids – oven cleaner is an example.
The second reason is more interesting – vinegar, Coca Cola, lemon juice and even water can all be considered as “acids”. It would be impossible (and pointless) to ban people from carrying these liquids.
So, what is an acid? The most common definition is that an acid is something that acts as a source of hydrogen ions (post 16.39). Acids were first defined in this way, in 1923, independently by Johannes Brønsted in Denmark and Thomas Lowry in England. At about the same time, the American Gilbert N Lewis gave a more general definition of an acid. Acids are sometimes called Brønsted-Lowry or Lewis acids, to make it clear which definition is being used. But, almost always, when you see the word “acid” it means a Brønsted-Lowry acid.
Hydrogen chloride is a gas made up of molecules in which a hydrogen atom is covalently to a chlorine atom (post 16.30). When it dissolves in water most of these covalent bonds break to give hydrogen (H+) and chloride (Cl–) ions (post 16.39). This chemical reaction can be represented by
HCl → H+ + Cl–
(see post 16.33). A solution of hydrogen chloride in acid is called hydrochloric acid.
When sulfur trioxide is dissolved in water, it forms sulfuric acid, the acid most commonly used in “acid attacks”. Sulfuric acid is often represented as H2SO4, as if it consisted of molecules. But, in water, most of these molecules would break down to give hydrogen (H+) ions and sulfate (SO42-) ions. (Complicated ions like SO42-, and the acetate ion in the picture below, are described in post 16.40).
Hydrochloric and sulfuric acids are examples of strong acids – acids in which all, or most, of the molecules that might exist break down to give hydrogen ions.
Acetic acid (in vinegar) and citric acid (in lemon juice) are examples of weak acids in which few of the molecules break up to give a hydrogen ion and a negatively charged ion (acetate for acetic acid and citrate for citric acid). In the case of acetic acid, the chemical reaction leading to the formation of hydrogen and acetate ions is shown below.
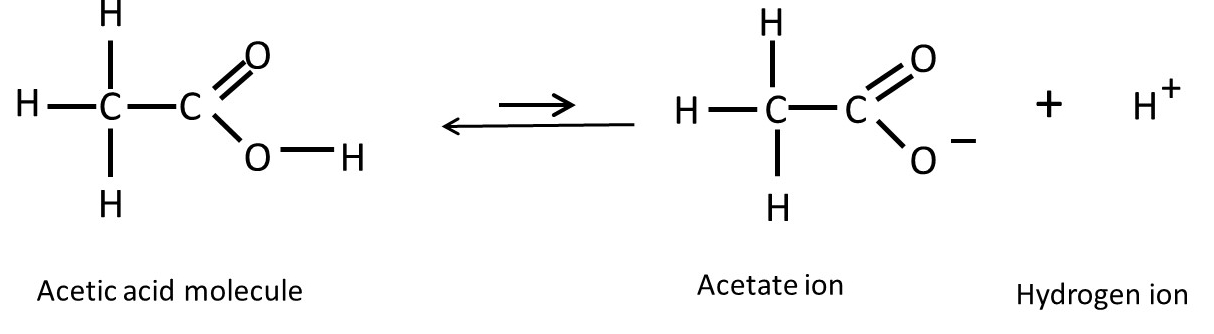
Note that the formula of acetic acid closely resembles the formulae of fatty acids in post 16.47. The double arrow shows that the reaction can occur in both directions. Acetic acid molecules can break down to give hydrogen and acetate ions. But hydrogen and acetate ions can also combine to form acetic acid molecules. Because acetic acid is a weak acid, few ions are produced – the reaction tends to proceed from right to left in the chemical equation (post 16.33) above; that’s why the right-to-left arrow if longer than the left-to-right arrow.
By now, you probably think that the difference between “strong” and “weak” acids is vague. You would be right! A better way to describe the strength of an acid is by the value of its acid dissociation constant. To explain this idea, let’s represent an acid molecule by HA; it’s an acid because
H-A →H+ + A–.
(Of course, the reaction could also go in the opposite direction). Now the acid dissociation constant of HA is defined by
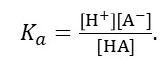
Here the [square brackets] mean the molarity of, for example, HA. Remember that molarity is a number (it doesn’t have the units of concentration – see post 17.48) so Ka is also a number with no units (see also post 17.41).
For example, let’s suppose that one in a thousand HA molecules break down into H+ and A– ions. The result will be that 999 HA molecules are left – the molecule that breaks down produces 1 H+ ion and 1 A– ion. Then
.
If you aren’t familiar with this way of writing numbers, or if you wonder why I haven’t given the answer as 1.001 × 10-3, see post 16.7.
For example, let’s suppose that one in a thousand HA molecules break down into H+ and A– ions. The result will be that 999 HA molecules are left – the molecule that breaks down produces 1 H+ ion and 1 A– ion ion. Then
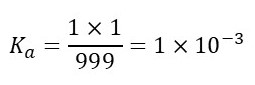 .
.
If you aren’t familiar with this way of writing numbers, or if you wonder why I haven’t given the answer as 1.001 × 10-3, see post 16.7.
For example, let’s suppose that one in a thousand HA molecules break down into H+ and A– ions. The result will be that 999 HA molecules are left – the molecule that breaks down produces 1 H+ ion and 1 A– ion. Then
If you aren’t familiar with this way of writing numbers, or if you wonder why I haven’t given the answer as 1.001 × 10-6, see post 16.7.
For example, the Ka values of hydrochloric acid, acetic acid and water are 1.0 × 10-2, 1.7 × 10-5 and 1.0 × 10-7, respectively; the weaker the acid, the smaller the value of Ka. The Ka value for water is important for other reasons (to be described in a later post) and is represented by Kw.
The main message of this post is that acids are not necessarily harmful – they are simply a source of hydrogen ions.
Related posts
17.48 Moles
16.40 Complicated ions
16.39 Ions
16.33 Chemical reactions
16.30 Molecules
16.27 Atoms
Follow-up posts
25.6 Henderson-Hasselbach equation
25.5 Organic acids
is the final formula for Ka given towards the end right … or should it read 10 to the power of -3 ?
LikeLike
Thank you for finding my mistake – I’ve now corrected it.
LikeLike