Before you read this, I suggest you read post 20.7.

For security reasons, there is a limit to the quantity of gels you can take on to an aeroplane. But do the passengers or security staff understand what a gel is? And what do the people responsible for the rules mean by the word “gel”?
A gel is a mixture of a polymer (post 20.7) and a liquid that, like a solid (post 16.37), is completely enclosed by its own free surfaces. So, unlike a fluid (post 16.37) it doesn’t flow. Jelly (“Jell-O” in the USA) is a flavoured gel of the polymer gelatine in water. It is made by adding hot water to gelatine. At this high temperature, the polymer chains have sufficient kinetic energy (see post 16.37) that the mixture of polymer and water is liquid. When the temperature drops, the polymers are less mobile and the water is trapped between the polymer chains. Gels in which the liquid is water, are called hydrogels.
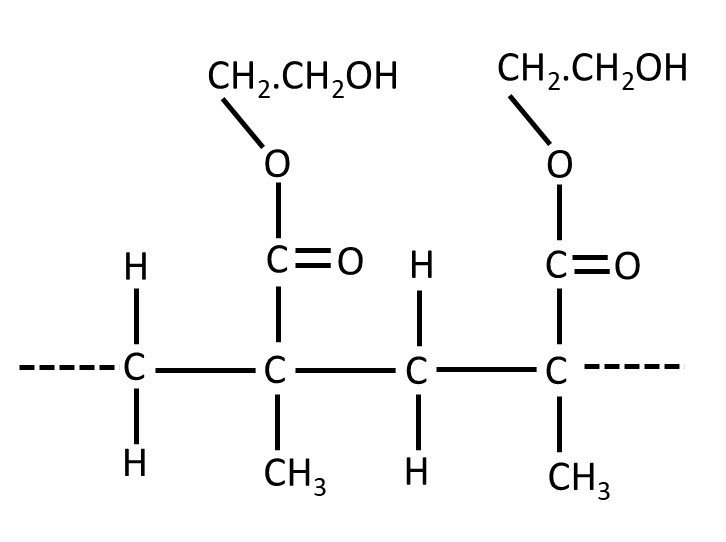
Since hydrogels can maintain their shape, they can be used to make soft contact lenses. Soft contact lenses are usually made of poly(2-hydroxyethyl methacrylate) (polyHEMA or pHEMA) whose formula is shown above. However, the pHEMA used to make contact lenses contains some cross-links (post 20.7). The cross-links create pores (like in a foam – post 20.5) that helps it to trap water. These foam-like pores are not totally enclosed (like in the closed-cell foam of post 20.5) but are like the cells in an open-cell foam, where communication is possible between the contents of adjacent cells, like trabecular bone (post 20.14).
There are other ways of linking polymer chains to create this pore effect besides the formation of covalent cross-links. For example, calcium (Ca2+) ions (post 16.39) can bind to groups on two different polymer chains that each have a single negative charge. Alginic acid, a polysaccharide (post 20.38) that can be extracted from brown seaweeds, forms gels that are be stabilised in this way. The effect is exploited in desserts (like instant crème caramel) that are made by adding milk (which contains calcium ions) to a powder to form a gel. Carageenans are gel-forming polysaccharides (post 20.38) that are widely used in the food industry. The sequence of monosaccharides in the polymer chain causes two chains to wrap around each other, forming coaxial helices, in some regions.
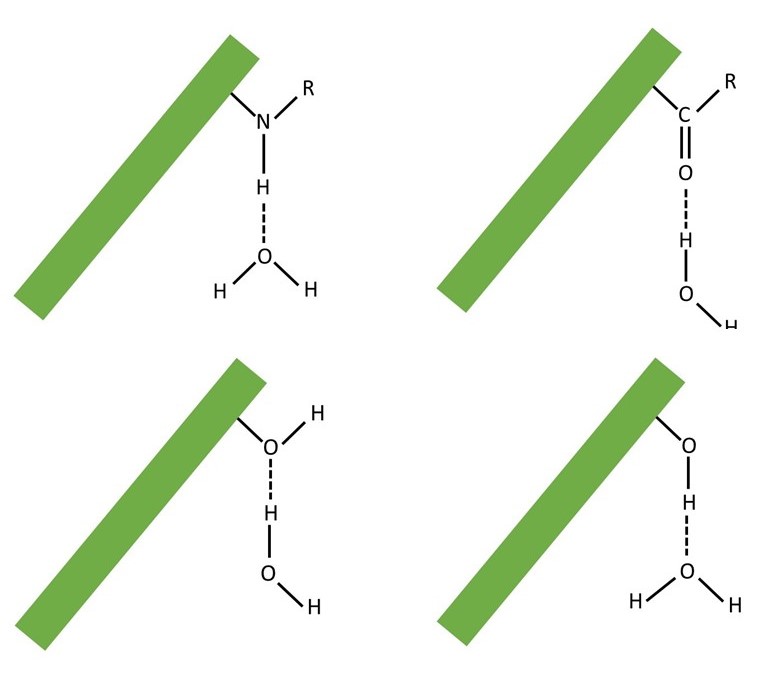
More importantly, there must be some force of attraction between water molecules and the polymer to prevent the water from flowing out of a hydrogel. Hydrogen bonds can form between water molecules (post 16.45). They can also form between water molecules and a variety of chemical groups that may occur along a polymer chain – some examples are shown below; in these examples, R represents any chemical group and a hydrogen bond is shown as a dashed line. These are examples of the hydrophilic regions of a molecule, described in post 16.48.
Many of the tissues in our body contain hydrogels. The cells (post 16.18) in our bodies are surrounded by a hydrogel called the extracellular matrix. (Don’t confuse these biological cells with the cells in a foam – post 17.14). Some tissues, like cartilage and the intervertebral discs in our spine, consist mostly of extracellular matrix. The hydrogel is mixed with fibres of collagen (post 20.14). The extracellular matrix of some tissues contains few collagen fibres and are soft materials that appear gelatinous. Examples are the vitreous humour that fills the inside of the eye and the nucleus pulposus (the inner region of the discs of the spine) of young people. In some tissues the extracellular matrix has a higher proportion of collagen and appears less gelatinous – but it is a hydrogel reinforced with fibres of collagen. Examples are articular cartilage (that coats the ends of bones in joints – like the knee) and the annulus fibrosus (the outer region of the discs in the spine), as well as the nucleus pulposus of older people.
So, to obey the regulations when we board a plane, we should really remove some parts from our bodies!
Related posts
20.7 Polymers
19.3 Real solutions
18.28 Applying the ideal gas equation to solutions
16.45 Water
16.37 Solids, liquids and gases
Follow-up posts