Before you read this, I suggest you read posts 22.4 and 22.21.
Post 23.1 was about x-ray diffraction by a crystal. If you learnt about this topic at school or university, you were probably taught Bragg’s law. We don’t need to know about this law to understand x-ray diffraction by a crystal; it uses a very different approach to the topic than the one I have used. But I thought it might be interesting to see how Bragg’s law provides an alternative explanation of x-ray diffraction.
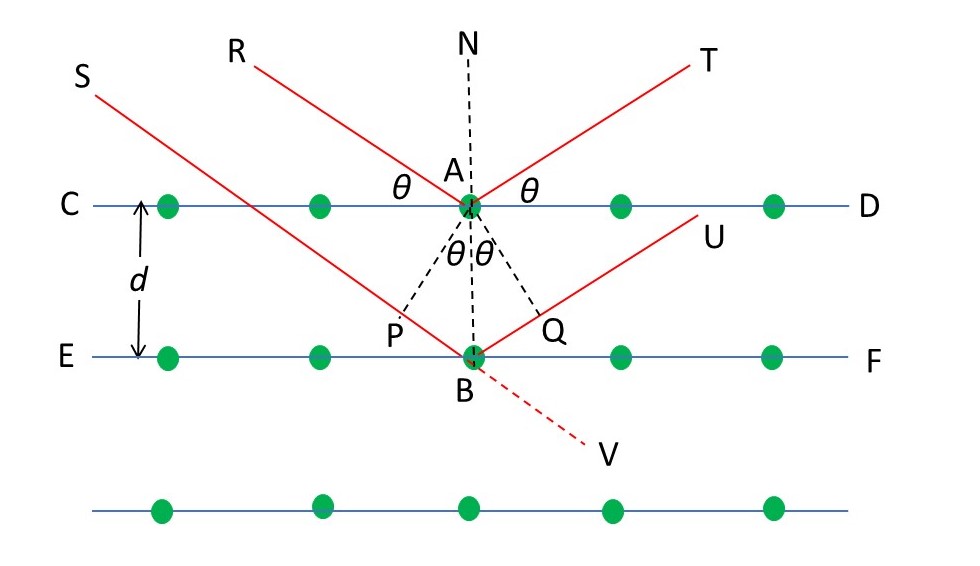
What is Bragg’s law? It is named after the British physicist (born in Australia) Lawrence Bragg (1890-1971) who formulated the geometry of diffraction by a crystal by supposing that the x-rays were reflected by planes of atoms. The picture above is a section through a crystal showing the positions of some of the atoms (coloured green). The blue lines (CD and EF) through the atoms show where the planes through them intersect the plane of the picture. The distance between the planes is d. RA and SB are rays incident on atoms A and B, respectively, that make an angle, θ, with the blue planes. The line BAN is perpendicular to the blue planes. Angles RBN and SBP are the angles of incidence for the rays. Angles NAT and NBU are the angles of reflection. According to the laws of reflection, the angles of incidence and the angles of reflection are both equal and have the value 90o – θ. Since NAB is perpendicular to the reflecting planes, angles TAD and UBF must be equal to θ. The lines AP and AQ are drawn perpendicular to the incident and reflected rays, respectively. Then angles CAP and DAQ are both equal to 90o – θ; as a result, angles PAB and QAB are both equal to θ.
If there is not to be destructive interference between waves reflected by the planes, the length PB + NQ must be equal to an integral number of wavelengths. If this were not the case, there would be a gradual shift in path lengths between reflected waves, as we move from one plane to the one below, and, eventually, there would be destructive interference. These ideas are explained in post 18.10. Since PB and NQ are both equal to dsinθ, we can write the condition for constructive interference as
2dsinθ = nλ (1).
Equation 1 is the way in which Bragg’s law is commonly stated.
In the picture above, the atoms lie along lines (in three dimensions, in planes) perpendicular to the blue lines. A line of atoms could be defined as a side of the unit cell of a crystal. So let’s define this direction as the c-axis direction – then c = d.
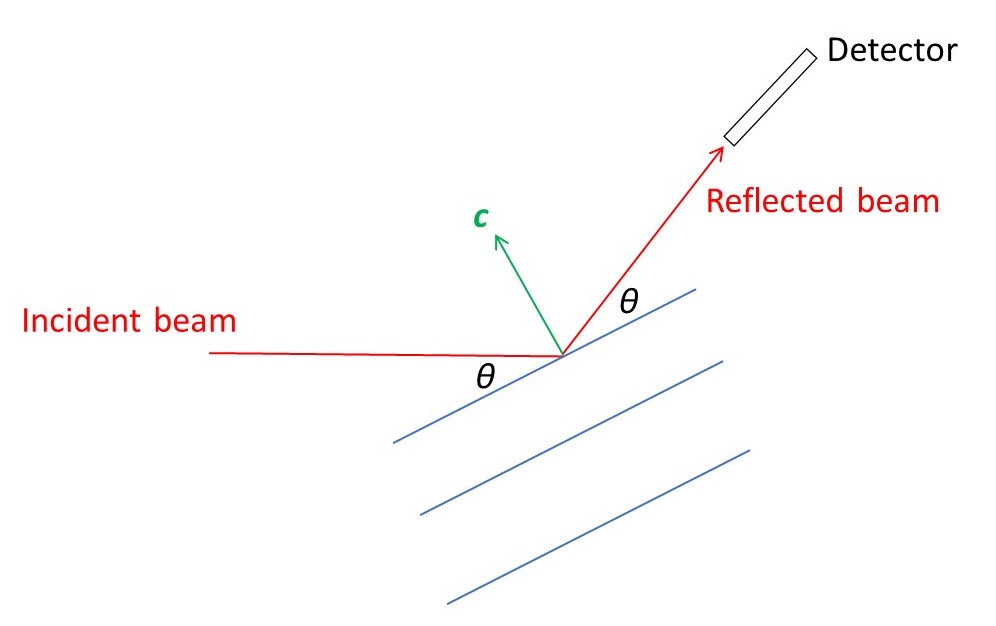
Now let’s suppose that we want to detect the values of θ that satisfy equation 1. We need to rotate the crystal about an axis perpendicular to the c-axis direction; for each rotation angle, we rotate a detector about this same axis to detect a reflection direction, such that the angle of incidence equals the angle of reflection, as shown in the picture below. How do we rotate the crystal about the c-axis direction? Remember that an ideal crystal is simply a stack of unit cells. So that any axis that occurs in the crystal shape is likely to correspond to the direction of a unit cell side. For example, the long axis of a needle-shaped crystal is likely to correspond to one of these directions that we could define as the c-axis.
There are several values of θ that satisfy equation 1, corresponding to n = 0, ±1, ±2, ±3….. . From equation 1, we can see, for example, that
sinθ-3 = -3λ/2d
sinθ-2 = -2λ/2d
sinθ-1 = –λ/2d
sinθ0 = 0
sinθ1 = λ/2d
sinθ2 = 2λ/2d
sinθ3 = 3λ/2d
Substituting this nomenclature into equation 1 gives
2(d/n)sinθn = λ (2).
If we had rotated about axes corresponding to the other unit cell directions, we would have observed different values of θn. To distinguish between these different sets of values for θn, we can write equation 2 as
2(c/l)sinθl = λ (3a)
2(b/k)sinθk = λ (3b)
2(a/h)sinθh = λ (3c).
Here h, k and l are n values corresponding to rotation about axis in the directions of the unit cell sides a, b and c, respectively.
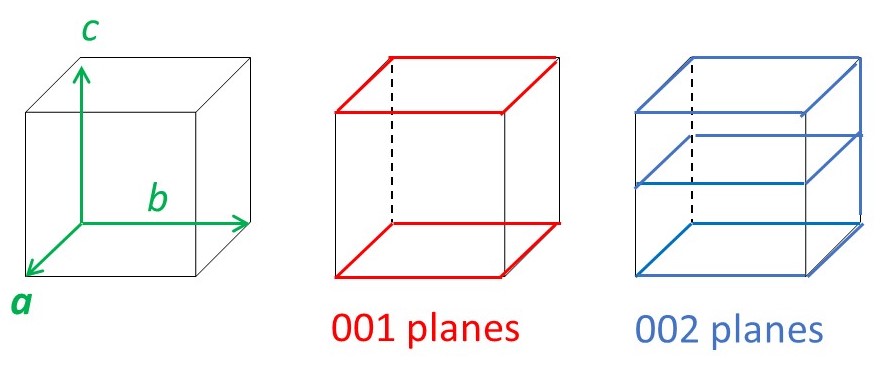
We have seen that the l = 1 reflection is from planes spaced a distance c apart that are perpendicular to the c-axis. When l = 2, we could interpret equation 3a as reflections from planes that are c/2 apart and, when l = 3, as reflections that are spaced c/3 apart. So we could write equation 3 as
2d00lsinθ00l = λ (4a)
2d0k0sinθ0k0 = λ (4b)
2dh00sinθh00 = λ (4c)
where d00l, d0k0 and dh00 are the spacings between planes generated by cutting the c, b and c axes, respectively, into 1/l, 1/k and 1/h parts, as shown in picture below.
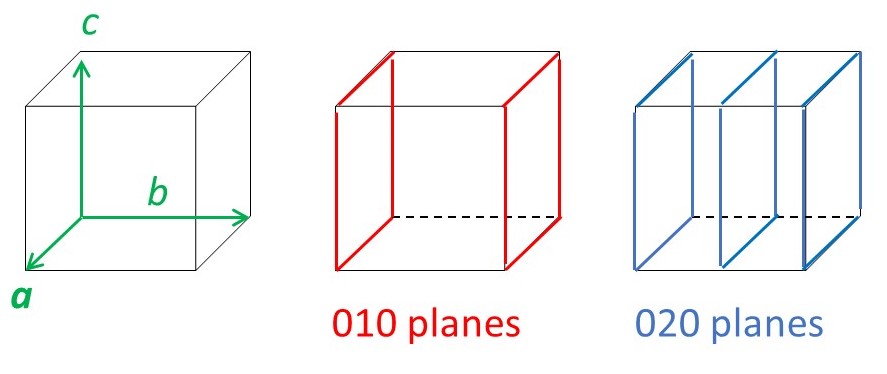
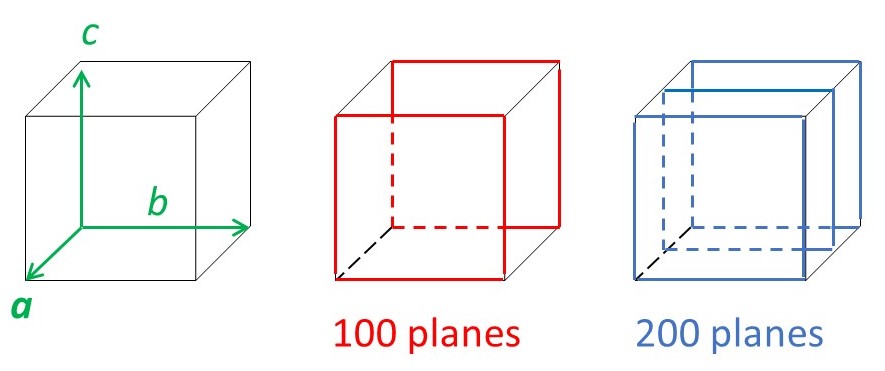
There are other planes of atoms in a crystal. The picture below shows some examples.
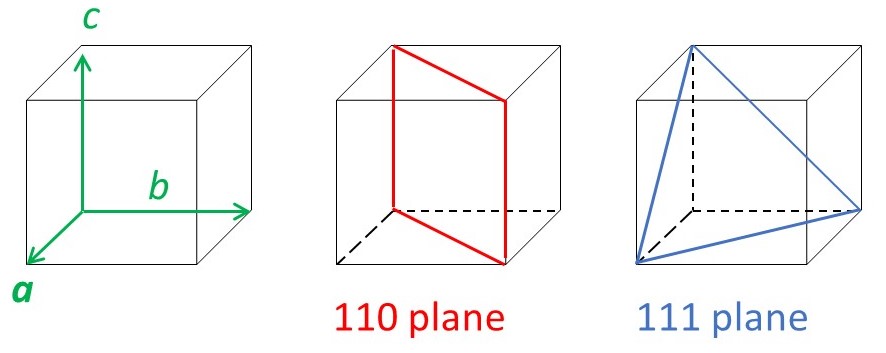
Now we can write equation 4 in the form
2 dhkl sinθhkl = λ (5)
where dhkl is the distance between the hkl planes. Equation 5 is another way of writing Bragg’s law. Note that to observe reflections that don’t have two of the integers h, k and l equal to zero, we must rotate about two axes.
So, Bragg’s law predicts the angles at which x-rays diffracted by a crystal will be observed. In post 23.1, I calculated the same results which ultimately were derived from the concept of K-space. The approach used here is much simpler and gives the same results (see appendix 1).
So why bother with K-space? The reason is that to use x-ray crystallography to determine the structure of molecules, which is its main application, we need to know something about F(h,k,l), the complex number that represents the wave reflected by the hkl plane in both amplitude and phase. Unfortunately, Bragg’s law tells us nothing about F(h,k,l) or how to use it.
Related posts
23.1 Observing x-ray diffraction by a crystal
22.24 Reciprocal lattice
22.23 K-space
22.22 Fourier transform of a one-dimensional lattice
22.21 An ideal crystal
22.14 X-ray diffraction
22.4 Reflection
Follow-up posts
Appendix 1
In post 23.1, we described x-ray diffraction using the scattering angle ϕ. In appendix 2, we see that θ = ϕ/2.
Also, in post 23.1, we used the concept of K-space to find a relationship between c and ϕ00l. Here we show that this relationship is identical to the predictions of Bragg’s law.
We saw (post 23.1) that
c* = (4π/λ)sin(ϕ001/2) = (4π/λ)sinθ001
where (for the case chosen in post 23.1) c* = 2π/c.
If you are really interested in this subject, the case considered was for a non-triclinic crystal with c as the unique axis; if you don’t understand this sentence – ignore it!
We can now write
sinθ001= (2π/c)(λ/4π)
or
2csinθ00l = λ.
Noting that c = d001, we have obtained a special case of equation 4a.
Appendix 2
In previous posts, I called the sum of the angle VBF and UBF, the scattering angle, ϕ. The appendix shows that ϕ = 2θ.
To understand this appendix, you need to look at the first picture of this post.
RA and SB are in the incident x-ray direction.
AT and BU are in the reflected x-ray direction, so the angle VBU is the scattering angle ϕ.
The angles APB and AQB were defined to be 90o.
Since the sum of the angles in a triangle is always 180o, the angles ABP and ABQ are both equal to (90o – θ).
Since NB is defined to be perpendicular to the blue planes, the angles PBE and UBF are both equal to 90o – (90o – θ) = θ.
B lies on the straight line PV. EV is also a straight line. So the angles VBF and PBE are equal. Since the angles PBE is equal to θ, the angles VBF is also equal to θ.
The scattering angle, VBU, is equal to the sum of the angles UBF and VBF and so is equal to 2θ.