
In post 16.37 we saw that the atoms or molecules or ions in a solid are packed together so that the solid is completely contained within its free surfaces – it forms an object that we can hold without it flowing through our fingers. In an ideal gas, the atoms or molecules are free to move, do not interact with each other and do not form any free surfaces.
We can explain the many of the properties of solids by considering that they consist of ideal crystals. Ideal gases and ideal crystals are models that help us to understand the properties of matter – they are not exact descriptions of reality.
What do I mean by an “ideal crystal”? In an ideal crystal the ions or atoms (alone or bonded to form molecules) are arranged in a regular pattern in three dimensions. The picture at the beginning of this post shows a simple two-dimensional representation
What are the limitations of the idea of a real crystal? The ideal crystal does not describe the structure of an amorphous solid and many solids (for example, metals) are polycrystalline – they consist of many small crystals packed together. But we can often assume that each little crystal in a polycrystalline solid is ideal. When the temperature is above 0 K, the atoms in our ideal crystal must have kinetic energy (see post 16.35). So we must think of the positions of the atoms in our picture as their mean positions of moving atoms that are simple harmonic oscillators. Real crystals contain defects that can be impurity atoms or regions of irregularity in the regular pattern. We need to think about defects to understand crystal growth, diffusion in solids, some of the mechanical properties of crystals and the properties of semiconductors. But we can understand many of the properties of real crystals by considering them to be ideal. Finally, real crystals have surfaces. The atoms at the surface cannot be surrounded by a regular, repeating three-dimensional pattern of atoms (if they were, the surface wouldn’t exist!). So an ideal crystal must be infinite. This isn’t a problem because our ideal crystal is a model – not an exact description of reality. But it is usually a good approximation to assume a crystal is infinite because the dimensions of most crystals are very much greater than the dimensions of atoms.
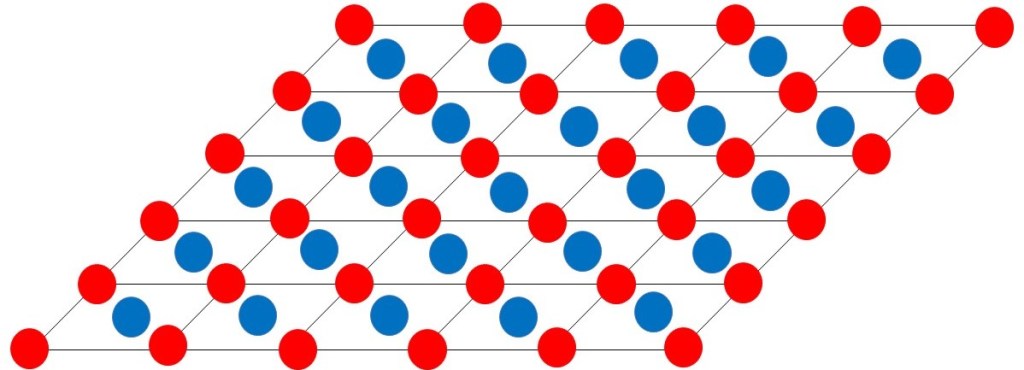
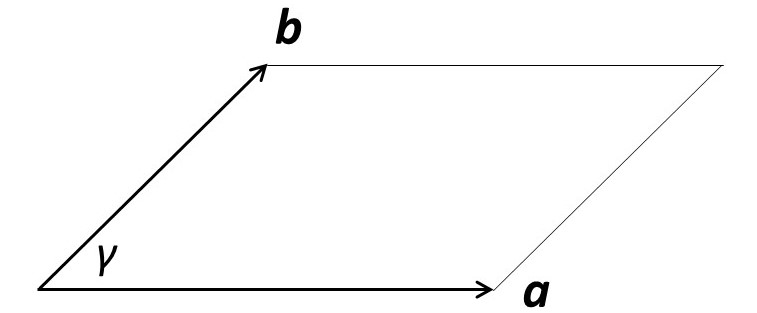
We can think of our ideal crystal as a lot of little boxes stacked together – as shown in the picture above. One of these little boxes is called a unit cell. The unit cell and its contents are the simplest structure that repeats itself in the crystal. Its dimensions and shape are defined by three unit cell vectors a, b and c – as shown, in two dimensions, in the picture below. In three dimensions, a, b and c form a right-handed coordinate system. The angle between b and c is usually called α, between a and c it is β and between a and b it is γ.
If we look at the points a = b = c = 0 for each unit cell, we see that they define a three-dimensional pattern of points – shown in two dimensions in the picture below; the positions of the points are marked by the centre of a cross. These points are called the crystal lattice or the Bravais lattice after the French physicist and statistician Auguste Bravais (1811-1863).

The, if we know a, b and c (the same thing as knowing a, b, c, α, β and γ) and the positions of all the atoms in a unit cell, we know the positions of all the atoms in our ideal crystal.
How do we define the positions of all the atoms in a unit cell? We define the position of the jth atom by the vector
rj = xa + yb + zc
where x, y and z are translations along a, b and c, respectively. Since the atom is within a unit vell the minimum values of x, y and z are 0 and they must be less than 1 – so they are called fractional unit cell coordinates.
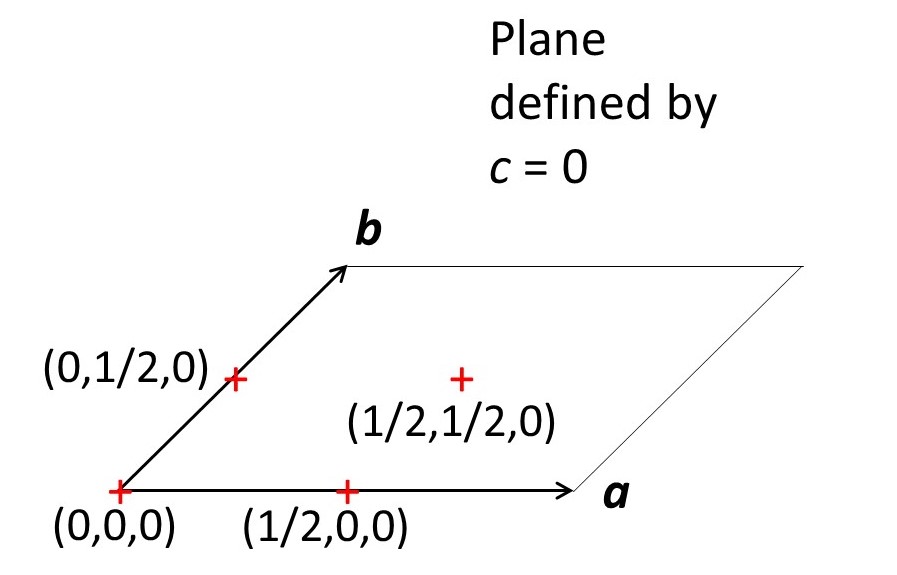
The picture above shows the positions of points (marked by the centres of red crosses) with fractional unit cell coordinates of (0,0,0), (1/2,0,0), (0,1/2,0) and (1/2,1/2,0). Fractional unit cell coordinates form a Cartesian coordinate system in which the axes are not necessarily 90o. In the picture above changing the value of y for an atom, in the b direction will change its x value, in the a direction, since γ ≠ 90o. So fractional unit cell coordinates do not form an orthogonal Cartesian coordinate system.
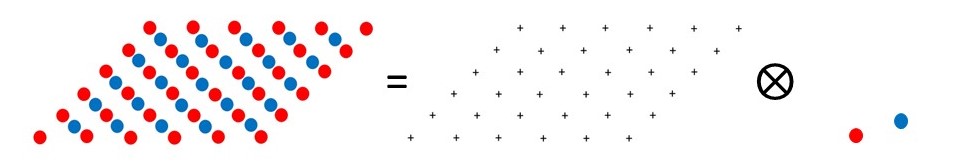
If you have read post 22.20, you will realise that the structure of an ideal crystal is the convolution of the unit cell contents with the lattice, as shown in the picture above.
Related posts
18.25 An ideal gas
16.37 Solids, liquids and gases
Follow-up posts
22.22 Fourier transform of a one-dimensional lattice
22.24 Reciprocal lattice
23.01 Observing x-ray diffraction by a crystal
23.02 Bragg’s law
23.03 X-ray diffraction by a crystal
23.04 X-ray crystallography