Many people believe two things about osteoporosis that are not true:
- It makes bones more brittle
- It is caused by loss of calcium from bones.
Osteoporosis makes bones less strong but not more brittle. Osteoporotic bone breaks at a lower stress than normal bone – so it’s less strong. But it does not appear to fracture more suddenly than normal bone – so it’s not more brittle (post 20.13).
To understand the reason for this, and to explain why osteoporosis is not caused by loss of calcium, we need to know more about the structure of bone. Cells (post 16.18) in bone are surrounded by a material that consists mainly of:
- A polymer (post 20.7) called collagen
- Filler particles (post 20.8) that are a form of calcium phosphate
- Water.
Calcium phosphates contain calcium ions (Ca2+, post 16.39) and phosphate ions (PO32-, post 16.40). The form of calcium phosphate in bone resembles imperfectly crystalline (post 16.37) hydroxyapatite that also contains hydroxide (OH–, post 16.40) ions. But the mineral in bone can also contain many other positively and negatively charged ions – so let’s call it bone mineral; we may shorten this to mineral when it is clear that we mean the mineral in bone. On its own, this mineral resembles a ceramic.
If the proportion of mineral is high, bone resembles a ceramic: if the proportion is low it resembles a polymeric material. Ceramics exhibit brittle fracture (you will see this if you break a plate or a cup) polymers exhibit ductile fracture and are tough materials (posts 20.10 and 20.13). So loss of calcium would occur because the bone lost mineral – it would then become more ductile not more brittle. Children’s bones contain less mineral than adult bone and so their fractures are ductile and not brittle; fracture of children’s bones is then usually an irreversible deformation called a greenstick fracture. (But a greenstick fracture can be reversed by external manipulation.)
So the two false ideas in the first sentence are contradictory. In order to understand why osteoporotic bone is less strong than normal bone, we need to know something about the structure of bones.

The picture above is represents the structure of a cross-section of a typical cylindrical bone; in this picture, bone material is coloured brown. This picture is not to scale; in reality, the little rods inside the hollow cylinder, and the spaces between them, are very small compared to the dimensions of the bone. The outer region consists of the material described above. The inner region consists of a network of little rods (made of the same material), called trabeculae (singular trabecula) that join in places, to form a solid foam (post 20.5). The spaces within this foam are filled with less dense, soft material called bone marrow or marrow. These two types or bone are called cortical bone (dense outer shell) and cancellous or trabecular bone (the solid foam). In this paragraph, I am explaining the structure of a bone (like a rib) and not the material that the bone is made of (which is what I was describing previously). A full description of a bone would then include other features like its nerves and blood vessels.
Bones need to be stiff – so that the forces exerted on them by muscles bend joints (like knees, elbows and ankles) and not the bones themselves (post 18.9). Now look at the diagram in post 20.9 that shows bending of the cross-section ABCD. The greater the width (AC and BD) the greater the tensile strain, ε, in AB, for a given bending angle. If the Young’s modulus of the bone material is E, the stress required to achieve this strain is σ = εE( post 20.2). The higher the stress, the stiffer the bone; here I’m thinking about the bone as an object not the bone material whose composition is described above. So if we want the bone to be stiff we want AC and BD to be as large as possible. This means that we want the cylindrical bone in the picture above to have a large radius.
If a bone had a large radius and was made entirely of cortical bone, it would have a large mass – because cortical bone is a dense material (post 16.44). If our bones had an unnecessarily high mass, we would waste energy moving our bodies (post 16.21). But if the centre of the bone consists of cancellous bone it will be almost as stiff but have a much lower mass, because cancellous bone is much less dense than cortical bone. The ability of a bone to be sufficiently stiff and strong (post 20.9) depends on it having the right proportions of cortical and cancellous bone.
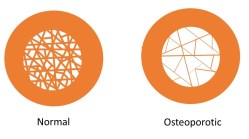
Osteoporosis is the loss of cancellous bone; this involves thinning and eventual loss of trabeculae. I will explain why this happens and why cancellous bone is lost more than cortical bone in the next post. The picture above is a diagram representing the differences between normal and osteoporotic bone. Because there is less bone material in osteoporotic bone, the bone fractures at a lower stress – which is why people with osteoporosis fracture their bones more easily.
I think many people believe that osteoporosis is calcium loss (or mineral loss) is because, when bone material is lost, mineral is lost too. Osteoporosis is then diagnosed by measuring the calcium content of the bone in order to measure how much bone material is lost. The technique used to measure the calcium content of bone is called dual energy x-ray absorptiometry (DEXA) and the result is called bone mineral density, although its not the same as the density defined in post 16.44.
Some people believe that drinking “Coca Cola” can cause osteoporosis because it is an acid (post 17.49) and so can dissolve bone mineral. But we have seen that this is not what happens in osteoporosis – so this belief is not true. There is another reason why it is not true that I will explain in a later post on homeostasis.
Related posts
20.13 Fracture
20.12 What happens in a pandemic?
20.9 Stiffness and strength
Follow-up posts
20.16 Homeostasis 1
20.15 Osteoporosis 2