Before you read this, I suggest you read post 16.33.

Stability is a more complicated idea than we often suppose. For example, a mechanical system can be stable but not in equilibrium because it is moving (with respect to an observer – see movement) on a stable path – it has dynamic stability. Similarly, a population of predators and prey, in an ecosystem, can be stable even though their numbers oscillate – see post 22.18.
In this post, I want to explore another complication involving the concept of stability – the stability of the products of a chemical reaction.
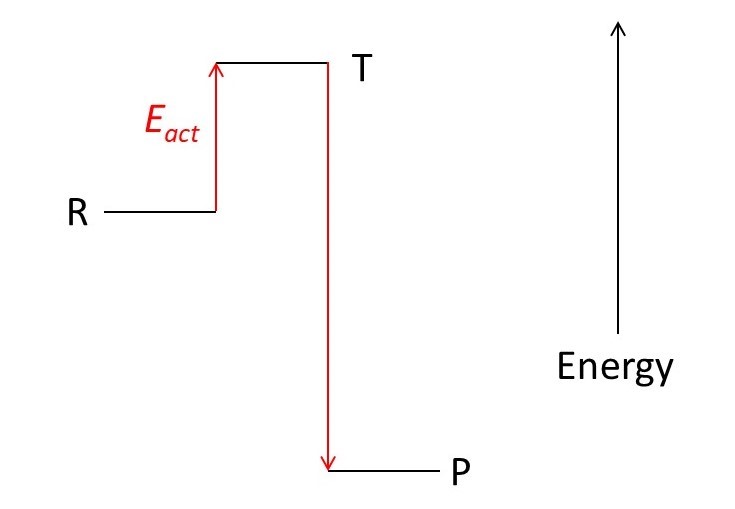
In the picture above reactants, R, are associated with an energy level. We can think of these reactants as being a collection of molecules. The products, P, of the reaction contain the same atoms as the reactants but arranged into different molecules. The reaction occurs because the products have a lower energy than the reactants. So the reaction lowers the energy of the system – in the same way that an object falls to lower its gravitational potential energy and so creates a more stable system.
Now I want to think a bit more about what we mean by “energy” in this context – remembering that learning about science is a process of diminishing deception. We are concerned with the energy available to do work, for example, if the products were to be used as fuel for a machine; we are not concerned with energy dissipated (see dissipation of energy) to increase the entropy of the system. So, in the context of this post, “energy” means “free energy” calculated using the Helmholtz function (if the reaction proceeds at constant volume and constant temperature), or, more usually, the Gibbs function (if the reaction proceeds under constant pressure at constant temperature).
To rearrange the atoms in reactant molecules to make product molecules, we first need to break chemical bonds in the reactant molecules. This requires energy, called the activation energy, Eact, usually in the form of heat. The high energy system created before the products can form is called the transition state, T. But it may be difficult to supply enough energy to form the transition state. For example, it may be difficult to increase the temperature high enough to supply enough heat.
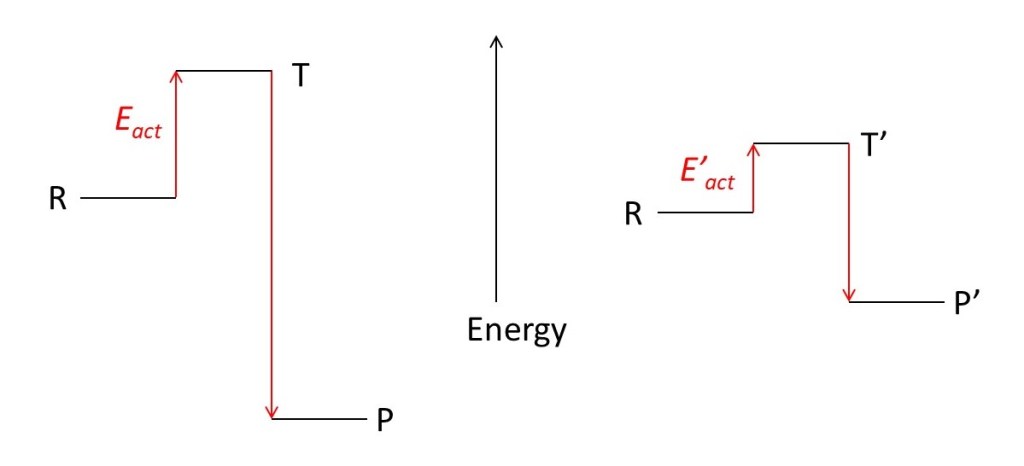
Now suppose there is a possibility of forming a different set of products, P’, that requires a lower energy transition state, T’. The energy of the reactants is independent of the products and so remains unchanged. If the energy of P’ is lower then R, the products of the reaction may be P’, instead of P, if E’act is much less than Eact. Here E’act is the energy difference between R and T’.
If P is the lowest possible energy state of the system, we say that P is thermodynamically stable.
Although P’ has a higher energy then P, it may be stable because insufficient R can gain state T in a realistic time. We then say that P’ is kinetically stable.
This may seem very abstract. Why haven’t I chosen a simple reaction as an example? The reason is that all he examples I could find involved very complicated chemical reaction. So you might suppose that the concept of kinetic stability applies only to very complicated systems. But that isn’t true.
Diamond and graphite are two different forms of the element carbon. But their atoms are bonded in different ways. In diamond, each carbon atom is joined to four other carbon atoms by single bonds. As a result, each carbon atom is at the centre of a tetrahedron with a carbon atom at each corner – in the same way that a carbon atom in a methane molecule is surrounded by four hydrogen atoms. But in diamond each carbon atom is surrounded by four carbon atoms, each of which is also surrounded by four carbon atoms, to form an extended three-dimensional network. So diamond is a very hard material that is used in cutting tools. In graphite the carbon atoms are joined by single and double bonds to form sheets. There are no chemical bonds between the sheets of carbon atoms – so they can easily slip past each other and carbon atoms can be rubbed from solid graphite. So graphite is used in pencils to make a mark on paper. These two forms of the same element with very different physical properties are called allotropes. We say that allotropy is the ability of some elements to exist in different forms – like diamond and graphite. Allotropes contain the same atoms but they are arranged differently in three dimensions.
You might be surprised to learn that graphite is the thermodynamically stable form of carbon. But diamond is kinetically stable. If you own a diamond ring – don’t be alarmed. The activation energy required to break all the C-C bonds in a diamond is so high that your diamond will never spontaneously convert into the thermodynamically more stable graphite.
Related posts