Before you read this, I suggest you read posts 16.29 and 16.30.
In post 16.30 we saw that the formation of molecules could be explained by atoms sharing pairs of electrons. This shared pair of electrons is called a chemical bond. If two atoms contribute one electron each, the bond is a covalent bond; if one atom contributes both electrons, the bond is a coordination bond.
This theory of the formation of molecules is called the valence bond theory. If you have learnt about this topic at school or university, this is probably the approach that was emphasised. It helps us to understand why some compounds exist but not others and enables chemists to plan how to make new compounds and to explain how they are formed.
But the valence bond theory can’t explain all the properties of molecules very successfully. For example, oxygen shows a weak form of magnetism called paramagnetism. Paramagnetism occurs when an atom or molecule has unpaired electrons; that is at least one orbital contains only one electron. If there is more than one unpaired electron, they spin in the same direction (see post 16.29). Since the electrons spin in the frame of reference of an observer, their charge creates a magnetic field (see post 16.25). According to the valence bond theory, oxygen exists as an O2 molecule in which all the electrons are paired, so there should be no overall electron spin because all electrons are paired and spinning in the opposite direction (see post 16.29). According to the valence bond theory, the only explanation for the paramagnetism of oxygen is that the two atoms are held together not by two bonds (O=O) but by one bond, leaving one unpaired electron on each oxygen atom (·O-O·). But why should the two electrons on each oxygen atom behave differently? Why should one form a covalent bond and the other not? It is difficult to think of an explanation that is not very contrived.
The molecular orbital theory takes a completely different approach that immediately predicts that oxygen will be paramagnetic. We have seen that atoms have atomic orbitals, corresponding to the allowed energy levels, in which there is a high probability of finding an electron with that energy (see post 16.29). The molecular orbital theory considers that molecules have molecular orbitals, in just the same way as atoms have atomic orbitals. But there is a practical problem. We can calculate atomic orbitals by solving an equation called the Schrödinger equation. But it is usually much too difficult to solve this equation directly for molecules – because they are more complicated, as a result of having more than one atomic nucleus. However, we can find solutions to the Schrödinger equation for molecules by assuming that their molecular orbitals are formed by combining atomic orbitals; this is called the linear combination of atomic orbitals (LCAO) approximation.
The diagram below then shows the allowed energy levels for a molecule consisting of two identical atoms. However, it is conventional to neglect electrons in the 1s level when forming molecular orbitals because they are close to the atomic nuclei and have lower energy than electrons in the 2s and 2p levels. I suspect that this may be a result of confusing the LCAO approximation with the valence bond theory – it certainly confused me when I first learnt about it! The diagram also shows how the molecular orbitals can be formed by the combination of the atomic orbitals of the two atoms (neglecting the 1s orbitals to be conventional and so getting the results shown in the textbooks). In the diagram I have just shown one 2p level for each atom; there are really three of them (2px, 2py and 2pz, as described in post 16.29), all corresponding to the same energy, so that, when they are combined in the LCAO approximation, they produce 6 molecular orbitals.
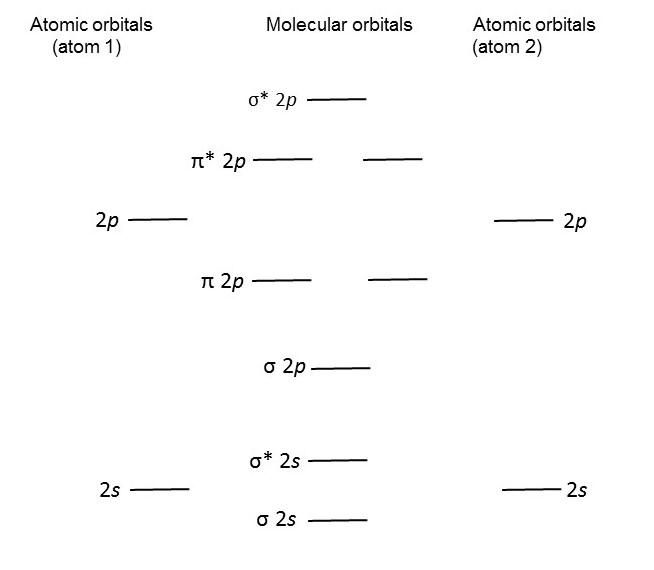
Now let’s think about an oxygen molecule. It has two atoms, each with eight electrons; so the oxygen molecule has 16 electrons. Of these 16 electrons, 4 are in the 1s level, so 12 are left to put into the molecular orbitals shown above. This happens in exactly the same way that atomic orbitals are filled by electrons in atoms (see post 16.29). Electrons go into the lowest energy levels first. Each atomic orbital can accommodate two electrons that spin in the opposite direction. When atomic orbitals correspond to the same energy (like the 2px, 2py and 2pz levels in atoms, see post 16.29), one electron goes into each and all spin in the same direction; when all contain one electron, a second electron can go in with the opposite spin.
When we put the 12 electrons into the allowed energy levels shown above, we get the result shown in the diagram below. In this diagram an arrow pointing upwards represents an electron with a spin in one direction; an arrow pointing downwards represents spin in the opposite direction.
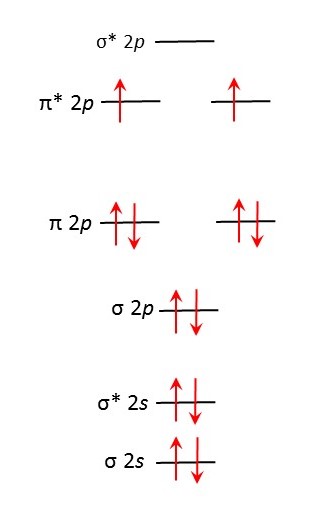
As a result, the oxygen molecule has 14 electrons that are paired with spins in opposite directions (including the 4 in the 1s levels) and two that spin in the same direction, so overall the electron spins do not cancel each other and the oxygen molecule is paramagnetic.
Why does oxygen form O2 molecules, according to the molecular orbital theory? Electrons in higher energy orbitals (σ* and π*) have higher energy levels than the atomic orbitals from which they were formed and so tend to disrupt formation of molecules; they are called anti-bonding molecular orbitals. But electrons in lower energy orbitals (σ and π) have lower energy levels than the atomic orbitals from which they were formed and so promote the formation of molecules; they are called bonding molecular orbitals. Oxygen has more electrons in bonding orbitals than in anti-bonding orbitals, so the O2 molecule is stable.
In more complicate molecules, molecular orbitals may extend over several atoms – unlike the bonds of valence bond theory that hold two atoms together; these extended molecular orbitals are called delocalised molecular orbitals. For example, a molecule of vitamin A (retinol) has π molecular orbitals that extend over 10 carbon atoms. (The valence bond theory can explain many of the properties of molecules with delocalised molecular orbitals by describing them as resonance hybrids, but explaining this would make this post much too long!) Delocalised molecular orbitals are very similar to the conduction bands that explain the electrical properties of metals and semi-conductors.
So there are two competing theories for the formation of molecules – the valence bond theory and the molecular orbital theory. I don’t think that one is better than the other. They are both very useful but for different purposes. The molecular orbital theory is simply too awkward to use to explain most of the questions that arise in chemistry (see the second paragraph in this post). Indeed one of the classic textbooks on chemical bonding, The Nature of the Chemical Bond by the Nobel-prizewinning chemist Linus Pauling (1901-1994), doesn’t mention the molecular orbital theory. But the molecular orbital theory immediately explains some things that are difficult to explain using the valence bond theory. It also has close similarities with the concept of the formation of conduction bands and so helps to bring together different areas of science. But one theory isn’t better than the other and it is not true that one theory is right and the other wrong!
Related posts
16.30 Molecules
16.29 Electrons in atoms
16.27 Atoms
16.2 Scientific laws
Follow-up posts
25.2 Sigma bonds and pi bonds
25.3 Benzene molecule