Before you read this, I suggest that you read posts 19.5 and 19.6.

In post 19.5, we saw that positive charges move spontaneously from hydrogen ions (H+) to zinc atoms because a zinc atom is at a lower electrical potential (post 17.44) than a hydrogen atom. As a result, a zinc atom is converted into a zinc ion, Zn2+ (post 16.39). But charges do not move spontaneously from H+ ions to copper (Cu) atoms because they are at a higher electrical potential. A simple electrical cell shows that the potential difference between zinc and copper is 1.10 V (post 19.6).
So, the Zn│Zn2+ system is at a lower potential than the H│H+ system that, in turn, is at a lower potential than the Cu│Cu2+ system . These three systems are examples of redox couples in which in which the first member of the couple is not charged but gains one or more charges to become the second member. Gaining charges in this way (by loss of electrons) is called oxidation (post 19.5). So the first member of the couple is the reduced form and the second term is the oxidised form (post 19.5). The potential difference between a redox couple and the H│H+ couple is called its redox potential or its standard electrode potential.
The table below gives some examples of redox potentials.
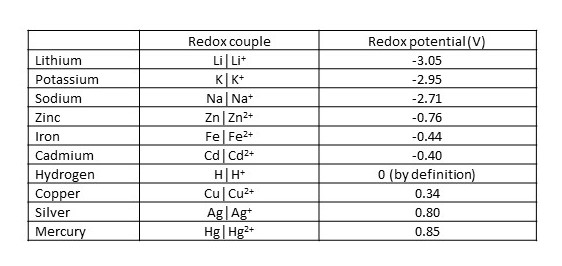
From this table, we can see that sodium, magnesium, zinc and ion can all displace hydrogen from acids, but that copper, silver and mercury cannot. Lithium, potassium and sodium have such low redox potentials that they can displace hydrogen from a very weak acid like water. You may have seen an experiment, when you were at school, in which sodium reacts violently with water to produce hydrogen.
You can also see that, if we make an electrical cell with silver and cadmium electrodes, the potential difference between these electrodes will be 0.80 – (-0.40) = 1.20 V.
So, to measure redox potentials, we need to make a hydrogen electrode. This isn’t easy because hydrogen is a gas. But it is possible to make a hydrogen electrode by bubbling hydrogen over platinum in a solution; hydrogen binds to the surface of the platinum to form a hydrogen electrode. Hydrogen electrodes are difficult to use. So normally a redox potential is measured relative to a more stable electrode (whose redox potential is known) and then the potential difference relative to a hydrogen electrode can be calculated
Redox potentials are useful in understanding other things. For example, many of the chemical reactions that occur in our bodies involve the transfer of charge from one molecule to another. Nicotinamide adenine dinucleotide, C21H27N7O14P2, is involved in many of these biochemical reactions in which charge is transferred. Because it is such a complicated molecule, it is often represented simply as NAD. However, it can also exist in the oxidised form NADH+, in which is bound to a hydrogen ion. The redox potential for the NAD│NADH+ couple is -0.32 V. So charge is spontaneously transferred to NAD from a redox couple with a higher redox potential. Remember that gaining a charge is equivalent to loosing an electron which is the way the process is usually described in biochemistry textbooks.
In summary, all these processes are examples of charge moving spontaneously from a high electrical potential to a low potential (post 17.44). This is the electrical analogue (post 18.24) of objects spontaneously falling from a position with high gravitational potential energy to a position with low gravitational potential energy (post 16.21).
Related posts
19.6 Cells and batteries
19.5 Oxidation and reduction
17.49 Acids
17.44 Amps, volts and ohms
16.39 Ions