Before you read this, I suggest that you read posts 16.2, 16.21 and 16.27
We can measure the energy of electrons in atoms from the wavelengths at which elements (see post 16.27) absorb or emit light. The Danish physicist Niels Bohr explained these results using the quantum theory (see post 16.2) in which electrons are only allowed to have certain “allowed” energy levels. It’s a bit like putting things on stairs; if each stair has a height h, and the things have a mass, m, they can only have potential energies (see post 16.21) of mgh. 2mgh, 3mgh and so on, where g is the gravitational field of the earth (see post 16.16), simply because they must be on a stair – they can’t be at some intermediate height.
The energy levels for electrons in atoms have names that are derived from early descriptions of the light absorbed by atoms and some additional features derived from the results of quantum mechanical calculations. We needn’t worry about where the names come from but the results, shown below, are very important. This diagram only shows the first few allowed energy levels, for simplicity.
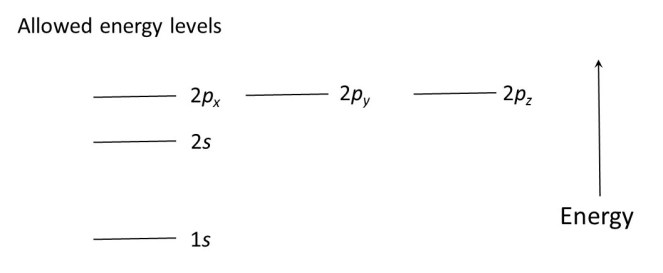
Notice that 2px, 2py and 2pz are three separate levels all corresponding to the same energy; the significance of this will become clear later.
In 1922 the German physicists Otto Stern and Walther Gerlach showed that electrons can spin in either a clockwise or an anti-clockwise direction. In 1927 another German physicist, Friedrich Hund, used the idea of electron spin to explain the occupation of the allowed energy levels in atoms.
The hydrogen atom has only 1 electron (Z = 1, see post 16.27). This goes in the lowest (1s) energy level. The helium atom has Z = 2; its two electrons can both go in the 1s level provided they spin in opposite directions. The lithium atom has Z = 3; two of its electrons go in the 1s level (provided they spin in opposite directions) and the third goes in the next lowest level – the 2s level. The diagram below shows the electrons in their allowed energy levels in a lithium atom; an upward pointing arrow denotes spin in one direction and a downward pointing arrow denotes spin in the other direction.
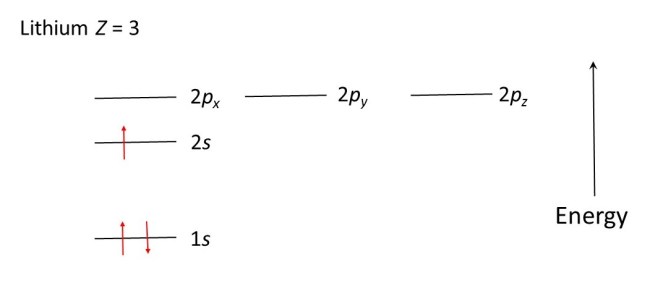
The beryllium atom (Z = 4) has two electrons in the 1s level (each spinning in the opposite direction) and two electrons in the 2s level (each spinning in the opposite direction). The boron atom (Z = 5) has two electrons in the 1s level (each spinning in the opposite direction), two electrons in the 2s level (each spinning in the opposite direction) and one electron in the 2px level, as shown in the diagram below.
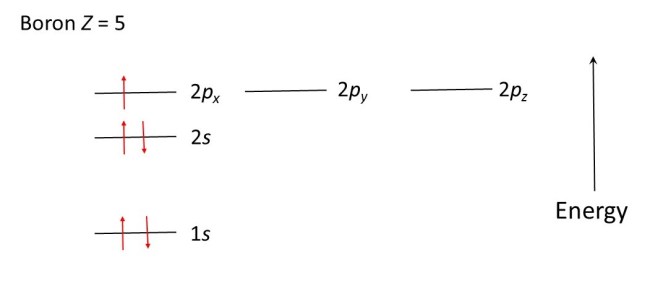
Have you asked yourself why the fifth electron of beryllium goes into the 2px level and not in the 2py or the 2pz level? The answer is that it doesn’t matter – there just happen to be three different 2p levels, corresponding to the same energy, that we arbitrarily label x, y and z.
Things get a bit more complicated when Z is greater than 5 because the 2px, 2py and 2pz levels all have the same energy. The carbon atom (Z = 6) has two electrons in the 1s level (each spinning in the opposite direction), two electrons in the 2s level (each spinning in the opposite direction), one electron in the 2px level and one electron in the 2py level; the electrons in the 2px and 2py levels have the same spin, as shown in the diagram below.

The nitrogen atom (Z = 7) has two electrons in the 1s level (each spinning in the opposite direction), two electrons in the 2s level (each spinning in the opposite direction), one electron in the 2px level, one electron in the 2py level and one electron in the 2pz; the electrons in the 2px, 2py and 2pz levels have the same spin, as shown in the diagram below.
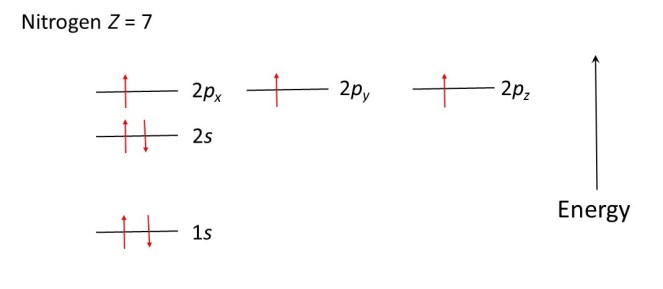 When Z is greater than 7, further electrons can share the 2px, 2py and 2pz levels, provided they spin in the opposite directions to the electrons that are already there. The diagrams below show how electrons occupy the energy levels in oxygen (Z = 8), fluorine (Z = 9) and neon (Z = 10) atoms.
When Z is greater than 7, further electrons can share the 2px, 2py and 2pz levels, provided they spin in the opposite directions to the electrons that are already there. The diagrams below show how electrons occupy the energy levels in oxygen (Z = 8), fluorine (Z = 9) and neon (Z = 10) atoms.
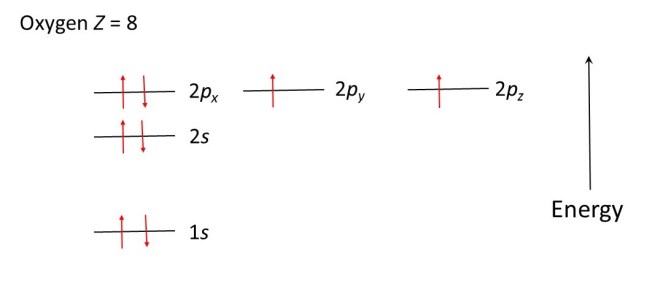
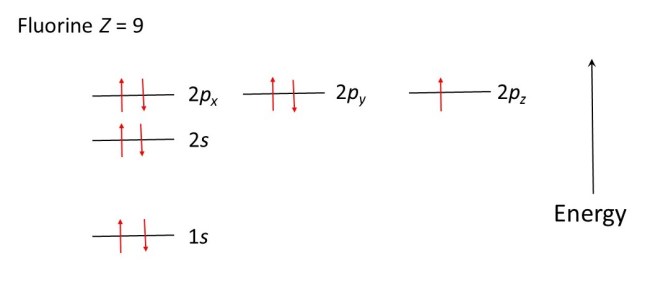
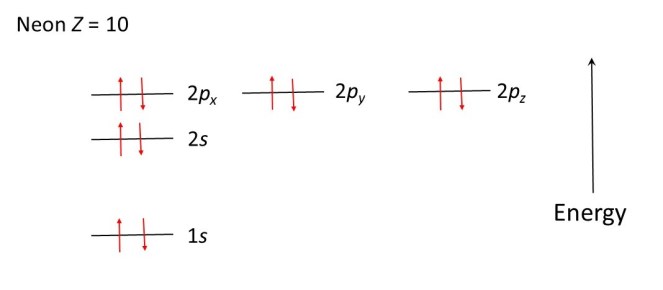 In much the same way as a ball will roll downhill, to lower its potential energy, electrons occupy the allowed energy levels in order to minimise the energy of the atom. The ball is in a more stable position when it has rolled downhill: the electron configurations shown in the diagrams above are the most stable. The most stable arrangement of its electrons is called the ground state of an atom. Absorption of light can lead the electrons to move to higher allowed energy levels; the atom is then said to be in an excited state.
In much the same way as a ball will roll downhill, to lower its potential energy, electrons occupy the allowed energy levels in order to minimise the energy of the atom. The ball is in a more stable position when it has rolled downhill: the electron configurations shown in the diagrams above are the most stable. The most stable arrangement of its electrons is called the ground state of an atom. Absorption of light can lead the electrons to move to higher allowed energy levels; the atom is then said to be in an excited state.
In post 16.2 we saw that, according to quantum theory, we can’t calculate the exact position of an electron. The best we can do is to calculate the probability of finding it in a given region of space; much like the 95% confidence levels in post 16.26. These regions of space in which there is a high probability of finding an electron with a given energy is called an atomic orbital. For example, the 2s atomic orbital is the region of space in which there is a high probability of finding an electron that is in the 2s energy level. So the familiar picture of the atom, like that in post 16.27, with electrons going round the nucleus, like plants round the sun, is really misleading. We don’t know exactly where the electrons are.
This is an example of Heisenberg’s uncertainty principle – that we can’t simultaneously know the exact energy and the exact position of an object. This principle doesn’t apply to everyday objects (like buses, for example – see post 16.2) because the uncertainties are so small that they are negligible. But it is very important for objects that are as small as electrons.
Related posts
16.27 Atoms
16.25 Electrical charge
16.2 Scientific laws
Follow-up posts
16.30 Molecules
16.31 Electrons in molecules
19.28 Solving Schrödinger’s equation
25.1 Hybrid orbitals