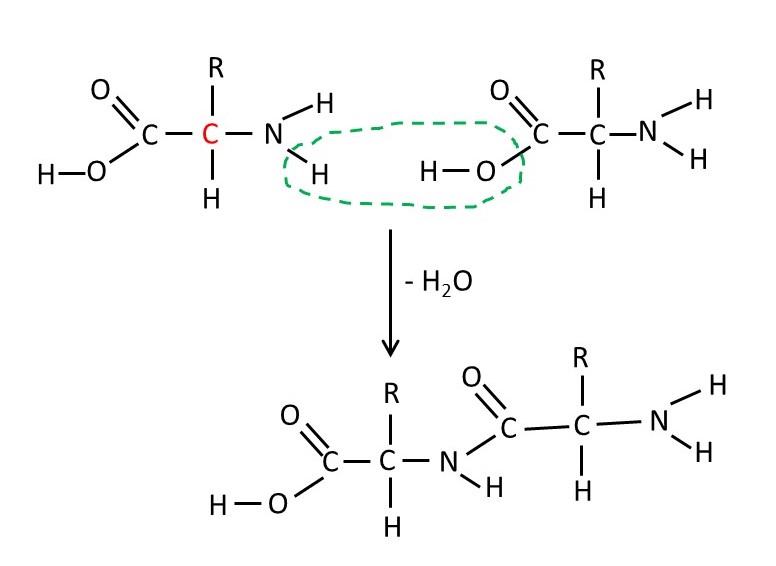
Proteins are condensation polymers of amino acids. The structural formula of an amino acid is shown at the top left-hand corner of the picture above, where R is any chemical group. (Two amino acids, proline and hydroxyproline, are slightly different). In post 20.27 we met alanine – the amino acid in which -R is the -CH3 group. Two amino acid molecules can join, with the loss of a water molecule, as shown above. When the process continues, the resulting condensation polymer is called a protein or (especially for synthetic polymers of amino acids) a polypeptide. The formula for part of a protein chain is shown below.
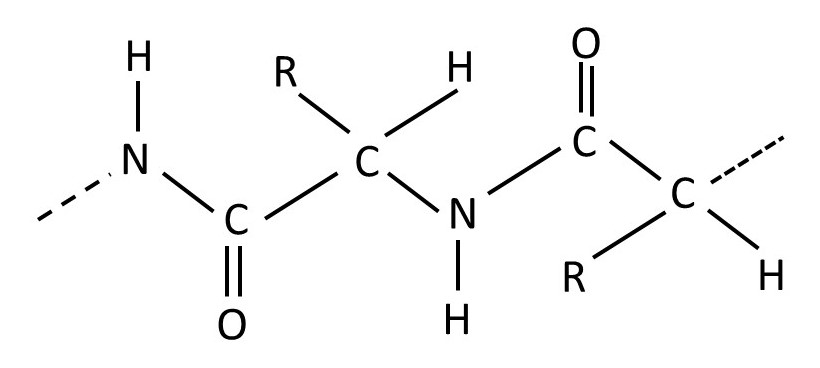
Unlike some polysaccharides, the polymer chains of proteins do not have branches.
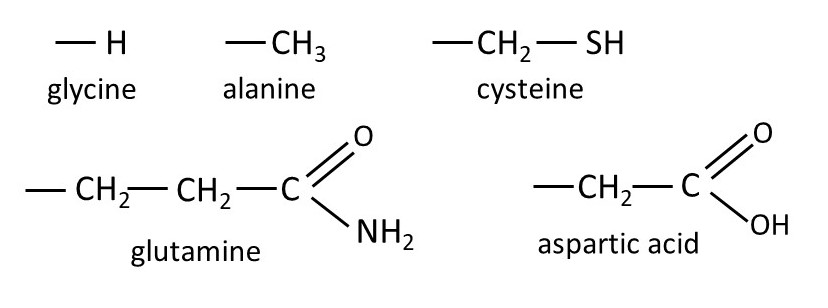
The picture above shows the R groups (side-chains) for several different amino acids. The bonds to the red carbon atom, in the amino acid shown at the top left-hand side of the first picture, point towards the corners of a tetrahedron – so amino acids are tetrahedral molecules. As a result, the amino acids (except for glycine) are chiral (they are not identical to their mirror images). Proteins contain only the L forms of amino acids – this is explained in post 20.27.
The sequence of amino acid residues determines the properties of the protein. For example, the glutamine side-chain tends to be positively charged because its -NH2 group can form a coordination bond with a hydrogen ion (from a water molecule), in the same way that the ammonium ion is formed. Similarly, the aspartic acid side-chain tends to be negatively charged because it can lose a hydrogen ion, in the same way that an acetic acid molecule can lose a hydrogen ion. By analogy with the properties of soap, the alanine side-chain is hydrophobic (the protein chain will fold to avoid the -CH3 group from contacting water). But the aspartic acid side-chain mixes readily with water (it is hydrophilic) and so the protein chain will fold so that it is in contact with water.
Cysteine side-chains can form cross-links between different polymer chains. Oxidation of two neighbouring -SH groups leads to the formation of water (H2O) and a covalent -S-S- bond.
Digestion of proteins involves breaking the polymer chains into individual amino acids that can be absorbed into the body by diffusion. We can then recombine the amino acids to make the proteins that our bodies require.
The proteins that we make have many different functions. Some proteins, for example collagen, form fibres that hold the body together. Muscles initiate and control movement; they are made of several different proteins. In some proteins, the polymer chain folds into a compact shape – they are called globular proteins. Enzymes (biological catalysts) are globular proteins; so are some hormones – for example, insulin that controls blood sugar levels. Haemoglobin, that occurs in red blood cells to transport oxygen around our bodies, is another globular protein.
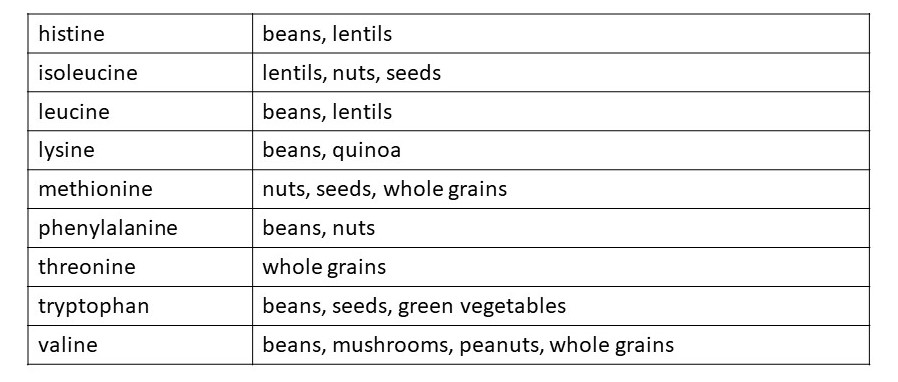
We can make some of the amino acids that we need. But there are nine amino acids which we cannot make and must obtain from our food – they are called essential amino acids. Similarly, we need to obtain the essential fatty acids that we can’t make from our food. The essential amino acids are listed in the table above.
Meat, fish and eggs are a useful source of proteins because they contain all the amino acids that our bodies need in roughly the right proportion. But plants contain proteins too. The table shows plant foods that are sources of the essential amino acids. You can see that we need to eat a mixture of plant-based foods to obtain all the essential amino acids. However, if we eat plenty of beans (that have been dried and then cooked), whole grains and green vegetables, we should be able to obtain all the amino acids we need.
In conclusion, proteins are essential for the structure of our bodies and for many of the chemical reactions that keep us alive. We eat proteins to obtain amino acids. Then we join these amino acids together to make the proteins that we need.
Related posts
21.2 Nucleic acids
20.40 Polysaccharides
20.38 Sugars
20.7 Polymers
16.47 Fats
Follow-up posts