Before you read this, I suggest you read post 19.24.

The uncertainty principle is named after Werner Heisenberg (picture above), the German physicist who suggested it in 1927. Heisenberg was awarded the Nobel prize in Physics in 1932. He was the first director of the Max Planck Institute for Physics; the Max Planck Institutes are the leading centres for scientific research in Germany.
To explain the uncertainty principle, I’m going to introduce a thought experiment. The idea is not to really do an experiment but to think what we believe would happen if we did. Thought experiments are commonly associated with the work of Einstein although “thought experiment” is an English translation of the term Gedakenexperiment introduced by the Austrian physicist Ernst Mach in 1897. But thought experiments were used by the Scottish physicist James Clerk Maxwell before the idea was described by Mach. Mach’s name is remembered by the Mach number that is associated with supersonic flight.
The uncertainty principle arises because an electron, or any other very small particle, can behave as either a particle or a wave (posts 19.24 and 19.25).
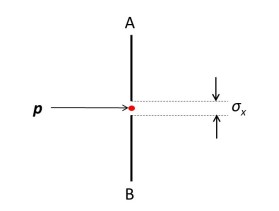
Let’s think about an electron moving in a straight line with momentum (defined as a vector quantity in post 17.39) p. (To find out more about vectors see posts 17.2 and 17.3.) We want to know its position when it passes through the plane AB in the picture above. To do this we place a screen with a hole in it in this plane. If the electron passes through the screen, it must be somewhere within the hole. To find its position more precisely, we could decrease the size σx of the hole.
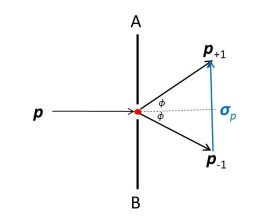
But if the size of the hole is comparable to the wavelength of the electron, we will see diffraction effects (post 19.20). Then the electron will be scattered in different directions, as shown in the picture above. If φ is the angle that encloses most of the scatter, the momentum of the electron can range from p-1 to p+1. Now there is an imprecision
σp = p+1 – p-1
in the value of the momentum. The vector σp, defined by this equation, is shown in the picture above.
Note that, for any momentum p,
p2 = p.p = (mv).(mv) = m2v2 = 2m(mv2/2) = 2mK.
Here m is the mass of the electron, v is its velocity and K is its kinetic energy (post 16.21). Dot products of vectors, like p.p, are defined in appendix 2 of post 17.13. So any imprecision in momentum, means an imprecision in energy.
The result of our thought experiment is an imprecision σx in the position of the electron and an imprecision σp in the modulus of its momentum and, hence, in its energy.
How great is this imprecision? We can’t use our thought experiment to get an exact result, but we can get a rough idea. According to post 19.20, φ, σx and the wavelength, λ, of the electron are related by
σxsinφ = nλ
where n is an integer. Taking the case where n = 1, we get the result that
sinφ ≈ λ/σx.
This is an approximation since n could be another integer. The picture above shows that
σp = 2psinφ ≈ 2pλ/σx.
The first stage arises from the definition of the sine of an angle (appendix 3 of post 16.50) and by representing the modulus of p+1 and p-1 by p. Multiplying both sides of this result by σx and noting that p = h/λ (post 19.24), we get the result that
σxσp ≈ 2h.
So, if we multiply the imprecision in the position of the electron by the imprecision in its momentum, the result is twice Planck’s constant, h, as defined in post 19.19.
Heisenberg’s uncertainty principle is important if we want to understand the motion of very small particles, like electrons. But for everyday objects the effect is negligible. In post 19.25, we saw that the momentum of a bullet is about 8 kg.m.s-1 but the value of Planck’s constant is only 6.626 × 10-34 m2.kg.s-1, so the value of σxσp is negligible compared with the magnitude of the momentum and distances involved in its motion.
Related posts
19.25 Wave-particle duality
19.24 Electron waves
19.23 Photoelectric effect and photons