Before you read this post, I suggest that you read post 16.5.
At the centre of every atom there is a dense core called its nucleus. However, some nuclei are unstable and spontaneously disintegrate to form a more stable nucleus, giving rise to a different kind of atom. This process is called radioactive decay.
We can’t be sure when any given atom will decay. But in a reasonable lump of the stuff that is made up of these atoms there will be about 1 000 000 000 000 000 000 000 000 000 of them.
Let’s stop and think about how big this number is. A second is a very short period of time but this number of seconds is several billion times the age of the planet earth! In this same time period you could wander around the whole universe (all the galaxies, stars, planets and the space in between) at a comfortable walking speed.
That’s a lot of atoms! We can make statements about the behaviour of such large numbers that are so highly probable that, for all practical purpose, they are certain. So, if any atom can decay randomly at any time, there then will be some time period, t, during which half of them will have decayed. This period is called the half–life of the stuff. Let’s suppose that we started with a number, n, of these atoms. Then after a time period, t, we will be left with n divided by 2; this means we have n/2 of them.
We still have a huge number of our original atoms left. So we can apply exactly the same argument to them. At the end of the next time period t, that is after a total time of t + t = 2t a half of the remaining n/2 atoms will have decayed, leaving ½ × n/2 = n/4. We can keep applying this argument to successive half-lives, as shown in the table below.

This is a lot like exponential growth (Post 16.5) except that now the number of atoms is halving (instead of doubling) in each time period t. It is an example of exponential decay. The result is that initially the number of decaying atoms is very large but it progressively decreases so that after eight half-lives only 1/256th of the original atoms are left. This can also been seen in the graph where the number of atoms (as a fraction of the original number) is plotted against time (expressed as multiples of the half-life).
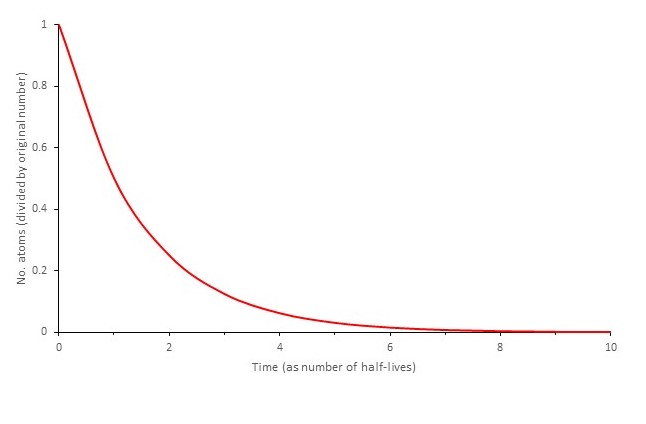
Since the number of atoms left keeps progressively halving it looks as if they will never all decay.
This is very like a paradox identified by the Greek philosopher Xeno about 2 500 years ago. Xeno pointed out that if you walk from A to B, you first walk a half the way, then a quarter of the way, then an eighth of the way and so on, with the result that you never actually arrive at B. At each stage the distance is halved but never becomes zero. Xeno’s paradox leads to all sorts of interesting mathematical and philosophical questions. But let’s consider the practical implications. Suppose A and B are 1 km apart. How do we know that you have arrived at B? We have to measure the distance between you and B; when it is zero you have arrived. So whether or not you have arrived depends on the precision with which you can measure this distance. By the eleventh halving of the way you are less than 1 mm from your destination. It’s very difficult to measure your distance from a place with this precision so, according to any reasonable measurement, you have arrived.
It often happens that the statements we can make are limited by the precision of the measurements we can make.
It’s much the same with radioactive decay. After a very large half-lives have elapsed, it would be impossible to detect whether there were any of the original atoms left.
The theory of radioactive decay illustrates another general point. The assumption that half the number of atoms will decay in one half-life is based on probability and works for a large number of atoms. But remember that we don’t know when a given atom will decay – it could be at any time. So when the number of unstable atoms is small, we don’t know anything about when they will decay. When stuff contains a very large number of atoms, we can make statements about its behaviour based on probability – but we can’t do this for small numbers of atoms. Many ideas in science are based on probability – so we can be sure about how large numbers of things will behave but can’t tell much about smaller numbers of them.
Related previous posts
Follow-up posts
16.27 Atoms
18.15 More about exponential growth