Before you read this, I suggest you read posts 18.1 and 18.3.

To calculate pH values, we need to understand more about the pH scale, for measuring acidity and alkalinity, that was in introduced in post 18.1. The definition of pH is that
pH = – log10[H+].
In this definition, [H+] represents the molar concentration of hydrogen ions (see posts 17.48 and 17.49) and “log10” means the logarithm to the base 10 (see post 18.3).
If we have a mole (post 17.48) of water, nearly all of it is present as H2O molecules but 10-7 moles of them break down to give 10-7 moles of H+ ions and 10-7 moles of OH– ions (post 17.49). Since there are equal numbers of the H+ ions that make the liquid acid (post 17.49) and the OH– ions that make it alkaline (post 17.50), water is neutral. From the definition of pH, the pH of water is then equal to
-log10(10-7) = 7,
as explained in post 18.3. This result explains why a neutral solution has a pH value of 7 (post 18.1).
If we dissolve the gas hydrogen chloride (HCl) in water, all its molecules break down to give equal numbers of H+ and Cl– ions. So the pH of 0.10 M HCl (see post 17.48 for an explanation of “0.1 M”) is
-log10(0.1) =-log10(10-1) = 1.0.
This result explains why the pH values of acids are positive numbers whose values are less than 7.
We saw, in post 17.49, that sulfuric acid is usually represented by the chemical formula H2SO4; but it is really a mixture of two H+ ions for every SO42- ion. So 0.10 M H2SO4 has [H+] = 2 ×0.1 = 0.2. So its pH is
-log10(0.2) = 0.70.
You can calculate -log10(0.2) using a spreadsheet (like Excel) or using your calculator.
However, when some acid molecules dissolve in water, not all the molecules break down to give H+ ions (acetic acid, post 17.49, is an example). Calculating the pH values of these acids is a bit more complicated and is explained in appendix 1.
How can we calculate the pH of an alkaline solution? Sodium hydroxide consists of equal numbers of sodium (Na+) and hydroxide(OH–) ions, so sodium hydroxide solutions are alkaline (post 17.50). But what is the molarity of H+ ions in 0.10 M sodium hydroxide solution? We have seen that, in water,
[H+] × [OH–] = 10-7 × 10-7 = 10-14.
Sodium hydroxide consists of sodium and hydroxide ions in water, so in 0.10 M sodium hydroxide solution
[H+] × 0.1 = 10-14 so that [H+] = 10-13.
From the definition of pH, the pH of 0.10 M sodium hydroxide solution is 13. You may also notice that no solution can have a pH value of greater than 14.
Ammonia (NH3) molecules can dissolve in water to form ammonium hydroxide, a mixture of equal numbers of ammonium (NH4+) and OH– ions (post 16.40). But not all the NH3 molecules form ions. So calculating the pH of 0.10 M ammonium hydroxide solution is more complicated than calculating the pH of 0.10 M sodium hydroxide solution and is explained in appendix 2.
More detail on calculating pH values is given at: https://www.chem.purdue.edu/gchelp/howtosolveit/Equilibrium/Calculating_pHandpOH.htm. If you read this web page, remember that all the H+ ions dissolved in water combine with H2O molecules to give H3O+ (hydronium) ions (post 16.40). Also pKa is defined to be -log10(Ka) and pKb is defined to be -log10(Kb).
These calculations show why pH values have a maximum value of 14, neutral solutions have a pH value of 7 and acidic solutions have a value of less than 7.
Related posts
18.1 pH
17.50 Alkalis
17.49 Acids
17.48 Moles
Follow-up posts
25.6 Henderson-Hasselbach equation
Appendix 1: the pH of 0.10 M acetic acid
In post 17.49, we saw that a few acetic acid molecules broke down in water as shown below.

We could denote this schematically as
H-A →H+ + A–.
We also saw that the acid dissociation constant was defined by

For acetic acid, Ka = 1.78 × 10-5.
Suppose that 0.1 M acetic acid contains x moles of H+ ions. It must then contain x moles of A– ions and (0.1 – x) moles of HA molecules. So
1.78 × 10-5 = x2/(0.1 – x).
To find the value of x we have to solve a quadratic equation – one that contains both x and x2. Appendix 3 explains how to solve a quadratic equation. But because the value of Ka is so small we can make a simplification; x will then be much smaller than 0.1, so that 0.1 – x ≈ 0.1. Our equation then becomes
x2 = 1.78 × 10-5 × 0.1 = so that x = 0.00133
pH = -log10(0.00134) = 2.9.
Appendix 2: the pH of 0.01 M ammonium hydroxide solution
Some NH3 molecules take up an H+ ion from a water molecule, to form an NH4+ ion, leaving behind an OH– ion, when they dissolve in water (post 16.40).
According to post 17.50, the base dissociation constant for this reaction is

Experiments show that the value of Kb for ammonia is 1.77 × 10-5. Suppose that 0.1 M ammonia forms x moles of NH4+. It must then contain x moles of OH– and 0.1 – x moles of NH3. So
1.77 × 10-5 = x2/(0.1 – x).
As in appendix 1, we have a quadratic equation to solve but, because the value of Kb is so low, we can assume that 0.1 – x ≈ 0.1, with the result that
x2 = 1.78 × 10-5 × 0.1 = so that x = 0.00133.
We have seen that, for a solution in water,
[H+] × [OH–] = 10-14
and have calculated that the value of [OH–] is 0.00133. So
[H+] = 10-14/0.00133 = 7.52 × 10-12.
pH = -log10(7.52 × 10-12) = 11.
Appendix 3: solving quadratic equations
The equation

has two solutions given by

where the symbol + means “plus or minus”. Both these solutions are mathematically correct but often only one will be relevant to the problem you wish to solve. So you need to use your judgement when interpreting the mathematical result.
For example, let’s solve the equation
![]()
In this example, a = 1, b = 1 and c = -6. So

The final step (√25 = 5) arises because 5 × 5 = 25 and √25 is just another way of writing 251/2 (see post 18.2). The two solutions are
x = -1/2 + 5/2 = 4/2 = 2 and x = -1/2 – 5/2 = -6/2 = -3.
Can you think how to solve this equation without using the formula?
Below I prove what I have written in the first paragraph.
If
![]()
subtracting c from both sides gives
![]()
and dividing both sides by a gives

Now let’s add b2/4a2 to both sides to get

Now we need to notice that
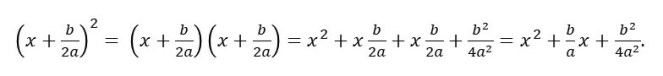
This means that equation 1 can be written as

Taking squares roots of both sides of this equation gives

The + sign arises because a positive number (like 9) can have a positive (+3) and a negative (-3) square roots because 3 × 3 = 9 and (-3) × (-3) = 9. Subtracting b/2a from both sides of the previous equation gives the required result.